Bupivacaine Hydrochloride
Bupivacaine Hydrochloride Specification
- Molecular Weight
- 324.88 g/mol
- EINECS No
- 238-119-8
- Melting Point
- Approx. 249-251C
- Boiling point
- Not applicable (decomposes)
- Storage
- Store in a cool, dry place, protected from light
- Heavy Metal (%)
- 0.002%
- Loss on Drying
- 0.5%
- Taste
- Bitter
- Smell
- Odorless
- Shelf Life
- 36 months
- Molecular Formula
- C18H28N2OCl
- Particle Size
- 20 micron
- Color
- White
- Poisonous
- Yes, toxic if overdosed
- HS Code
- 29420090
- Ph Level
- 4.0 6.0
- Solubility
- Freely soluble in water and alcohol
- Structural Formula
- C18H28N2OCl
- Medicine Name
- Bupivacaine Hydrochloride
- Chemical Name
- Bupivacaine Hydrochloride
- CAS No
- 14252-80-3
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Local anesthetic used for injection
- Purity(%)
- >99%
- Appearance
- White crystalline powder
- Physical Form
- Solid
- USP Reference
- Complies with USP/BP/EP Standards
- Residue on Ignition
- 0.1%
- Endotoxin Level
- Below specified pharmacopoeial limits
- Microbial Limits
- Complies with pharmacopeia requirements
- Packing Type
- HDPE Drums / Custom packaging available
- Manufacturing Method
- Synthetic process
- Assay
- >99% (HPLC)
- Identification Tests
- IR, TLC, HPLC methods confirmed
- Moisture Content
- 0.5%
Bupivacaine Hydrochloride Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Bupivacaine Hydrochloride
Bupivacaine Hydrochloride: Trusted by Medical Professionals
Used by anesthesiologists, surgeons, and healthcare practitioners, Bupivacaine Hydrochloride is a premium choice for surgical and pain management applications. Its customizable packaging and top-tier purity make it suitable for hospitals, clinics, and research facilities. Besides its primary use in local anesthetic injections, it also finds application in regional blocks and epidural pain relief, delivering reliable performance where precision is paramount.
Payment Terms & Logistics: Seamless Transactions and Verifiable Standards
Enjoy flexible payment terms designed for global clientele, with secure options to suit varying needs. Sample policy offers select samples upon request, ensuring complete confidence before procurement. All shipments are transported via certified goods transport networks-express shipping available for urgent requirements. Certified for international pharmacopoeia standards, the shipped goods arrive with verifiable documentation, guaranteeing peace of mind through every transaction.
FAQ's of Bupivacaine Hydrochloride:
Q: How is the quality of Bupivacaine Hydrochloride verified?
A: Quality is rigorously confirmed using identification tests such as IR, TLC, and HPLC, with each batch meeting USP/BP/EP standards and exceeding 99% purity.Q: What is the recommended storage method for Bupivacaine Hydrochloride?
A: It should be stored in a cool, dry place, protected from light, to maintain stability and prolong its 36-month shelf life.Q: Where is Bupivacaine Hydrochloride commonly used?
A: This product is primarily used in medical settings such as hospitals, clinics, and surgery centers for local anesthetic injections and regional anesthesia.Q: When can I request a sample of Bupivacaine Hydrochloride?
A: Samples can be requested at any stage of your procurement process. Our sample policy ensures you can evaluate product quality before placing a bulk order.Q: What are the main benefits of selecting customizable packaging for Bupivacaine Hydrochloride?
A: Customizable packaging allows you to choose specifications that best fit your storage, transport, and application requirements, ensuring convenience and product safety.Q: How is Bupivacaine Hydrochloride shipped and delivered?
A: Shipments are handled through reliable goods transport channels, with options for express shipping, and all deliveries are accompanied by thorough certifications and documentation.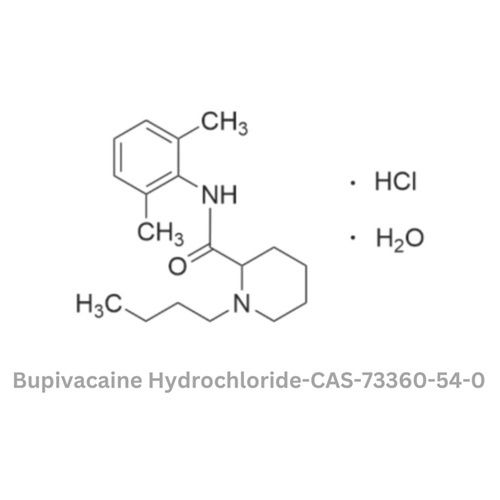

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
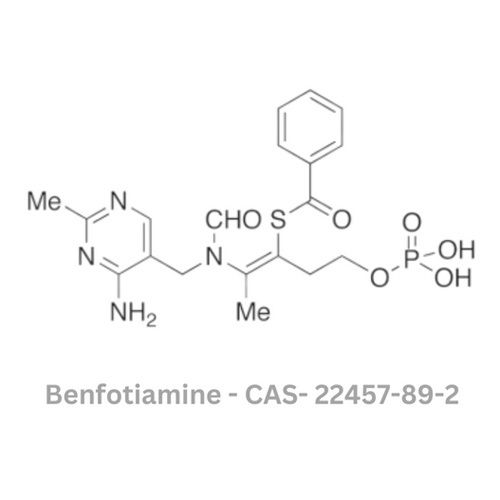
Benfotiamine API
Minimum Order Quantity : 120 Kilograms
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Taste : Other , Slightly Bitter
Smell : Other, Odorless
Usage : Used as a vitamin supplement, especially for diabetic neuropathy and other vitamin B1 deficiency conditions
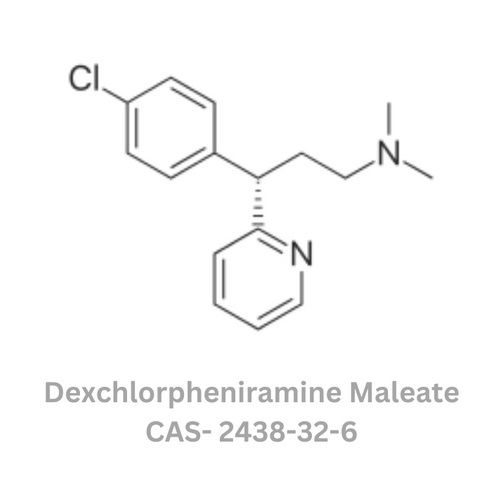
Dexchlorpheniramine Maleate
Grade : Other, Pharmaceutical Grade
Physical Form : Powder
Taste : Other , Slightly bitter
Smell : Other, Odorless
Usage : Used in the formulation of antiallergic and antihistamine medicines
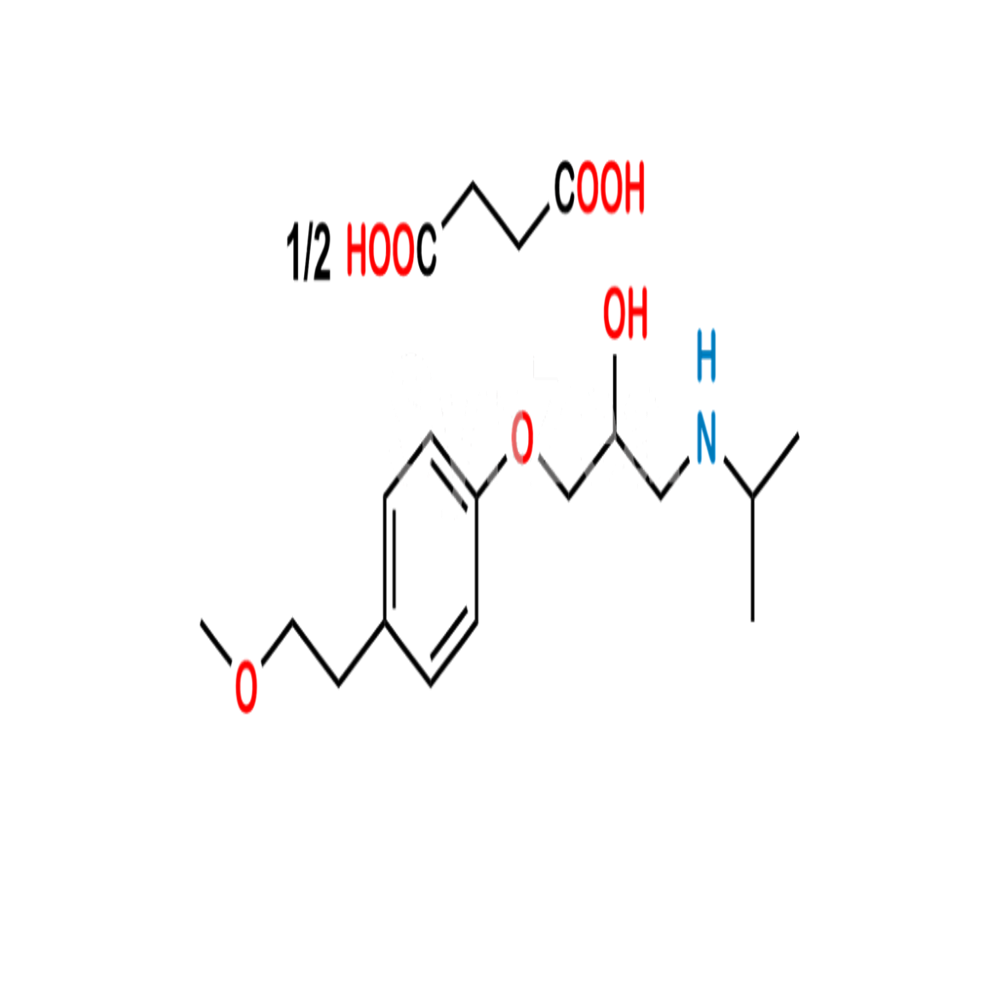
Metoprolol succinate IP/BP/EP/USP
Minimum Order Quantity : 100 Kilograms
Grade : Medicine Grade
Physical Form : Powder
Taste : Odorless
Smell : No Smell
Usage : A betablocker medicine used for the treatment of angina, high blood pressure and heart failure. Metoprolol succinate is also used for the treatment of migraine headache. It works by lowering the heart rate, blood pressure, and strain on the heart.
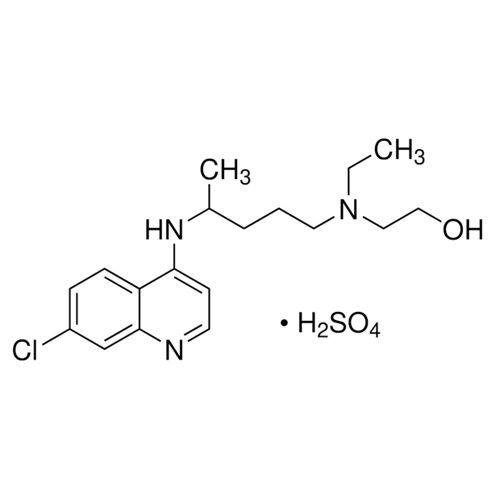
Hydroxychloroquine Sulfate API Powder
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Physical Form : Solid
Taste : Bitter
Smell : Stimulus
Usage : Commercial


 Send Inquiry
Send Inquiry