Rivaroxaban API
Rivaroxaban API Specification
- Melting Point
- 228-231 C
- Poisonous
- NO
- Loss on Drying
- <0.5%
- EINECS No
- ---
- Heavy Metal (%)
- <0.001%
- Particle Size
- D90 <10 microns
- HS Code
- 29420090
- Smell
- Odourless
- Solubility
- Slightly soluble in water; soluble in organic solvents
- Molecular Formula
- C19H18ClN3O5S
- Molecular Weight
- 435.88 g/mol
- Shelf Life
- 5 years if properly stored
- Storage
- Store in a cool, dry, well-ventilated area, away from direct sunlight
- Color
- White to off-white
- Structural Formula
- Shown as per chemical data sheet (structure not displayable in text)
- Medicine Name
- Rivaroxaban API
- Chemical Name
- Rivaroxaban
- CAS No
- 366789-02-8
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Anticoagulant, used for the prevention and treatment of blood clots
- Purity(%)
- >=99%
- Appearance
- White to off-white powder
- Physical Form
- Solid
- Assay
- >=99.0% (on dry basis)
- Microbial Limits
- Complies with pharmacopeial standards
- Stability
- Stable under recommended storage conditions
- Water Content (KF)
- <0.5%
- Identification
- Complies as per standard (IR, HPLC)
- Residual Solvent
- Within ICH guidelines
- Endotoxins
- Not detected
- Packaging
- Packed in double polyethylene bags placed in HDPE drum
- Related Substances
- Total impurities <0.2%
Rivaroxaban API Trade Information
- Minimum Order Quantity
- 100 Pieces
- Supply Ability
- 100 Pieces Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Rivaroxaban API
Distinctive Features and Quality of Rivaroxaban API
Rivaroxaban API stands apart for its exceptional purity (99%), rare low impurity content, and unassailable pharmaceutical grade. The material is a white to off-white, odourless solid with a reliable particle size at D90 <10 microns. Manufactured using valiant, state-of-the-art facilities, each batch undergoes robust identification (IR, HPLC), meets strict residual solvent and endotoxin guidelines, and guarantees compliance with pharmaceutical microbial limits. It is stabilized for extended shelf life, ensuring high efficacy and safety from production to patient.
Rivaroxaban API Samples: Dispatch, Policy, and Market Coverage
We offer Rivaroxaban API samples with a quick dispatch policy to expedite your evaluation process. Each sample is carefully handed over in secure packaging, guaranteeing product integrity. Our sample policy caters to both existing and prospective clients, ensuring fair access and clear guidelines. Samples are shipped securely, with all necessary documentation, to various destinations across the main domestic market, reflecting our commitment to timely delivery and reliable service.
FAQ's of Rivaroxaban API:
Q: How is the quality of Rivaroxaban API ensured?
A: Quality is unassailable, with each batch undergoing identification by IR and HPLC, compliance with ICH residual solvent guidelines, impurity content below 0.2%, and purity of 99%. Further, all products meet pharmacopeial microbial and endotoxin standards.Q: What is the recommended storage condition for Rivaroxaban API?
A: Rivaroxaban API should be stored in a cool, dry, well-ventilated area away from direct sunlight, ensuring stability and maintaining its 5-year shelf life.Q: When can customers expect sample dispatch after ordering?
A: Samples are dispatched quickly, typically within a short time frame after order confirmation, with secure shipping and thorough documentation for safety and traceability.Q: Where is the product mainly available and shipped from?
A: The Rivaroxaban API is manufactured and exported from India, with a strong presence in the main domestic markets and an established reputation as a reliable supplier.Q: What are the main benefits of using this Rivaroxaban API?
A: Benefits include a rare combination of high purity, low impurities, robust safety measures, and stability, making it a top-rated option for producing reliable and efficacious anticoagulant medications.Q: How is the product packaged to ensure its safety during transport?
A: The API is packed in double polyethylene bags placed within HDPE drums, providing layers of protection against moisture, contamination, and physical damage during transit.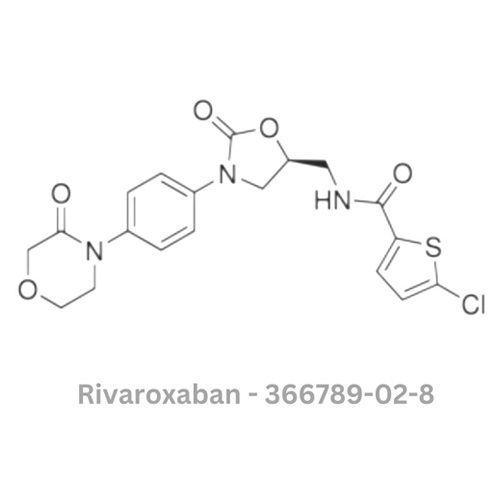

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
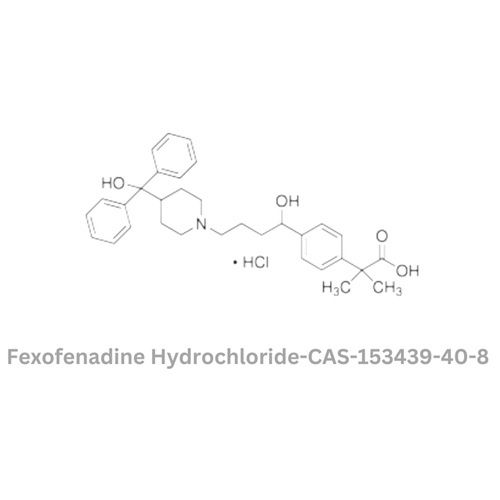
Fexofenadine Hydrochloride
Minimum Order Quantity : 100 Kilograms
Storage : Other, Store in a cool, dry, and wellventilated place, protected from light
Physical Form : Solid
Grade : Other, Pharmaceutical Grade
Smell : Other, Odorless
Type : Other, Antihistamine API
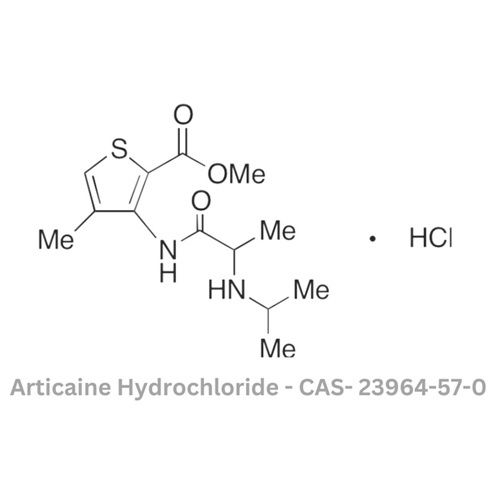
Articaine Hydrochloride
Minimum Order Quantity : 100 Bottles
Storage : Other, Store in cool, dry, and dark place
Physical Form : Solid
Grade : Other, Pharmaceutical Grade
Smell : Other, Odorless
Type : Other, Active Pharmaceutical Ingredient (API)
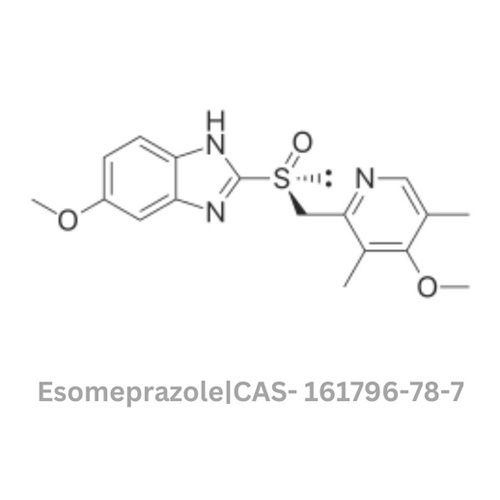
Esomeprazole API
Storage : Other, Store in a cool, dry place, protected from light and moisture
Physical Form : Powder
Grade : Other, Pharmaceutical Grade
Smell : Other, Odorless
Type : Other, Active Pharmaceutical Ingredient (API)
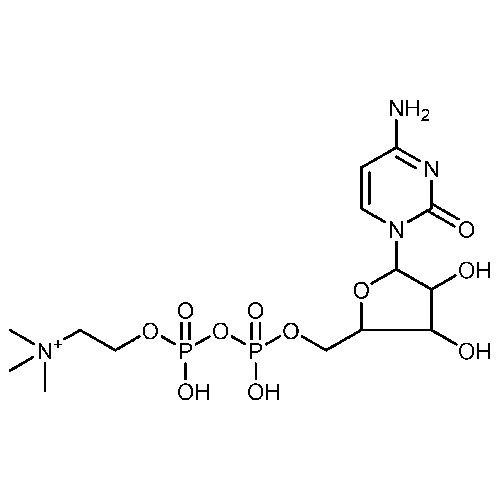
Citicoline Sodium API
Minimum Order Quantity : 100 Kilograms
Storage : Room Temperature
Physical Form : Powder
Grade : Industrial Grade
Smell : Stimulus
Type : Other, Citicoline Sodium API


 Send Inquiry
Send Inquiry