Acarbose API
Acarbose API Specification
- EINECS No
- 259-133-7
- Molecular Formula
- C25H43NO18
- Molecular Weight
- 645.6 g/mol
- Melting Point
- 167 - 170C
- HS Code
- 29400090
- Structural Formula
- C25H43NO18
- Boiling point
- Not applicable (decomposes)
- Medicine Name
- Acarbose API
- Chemical Name
- Acarbose
- CAS No
- 56180-94-0
- Type
- Antidiabetic Agent
- Grade
- Pharmaceutical Grade
- Usage
- Used in the treatment of type 2 diabetes mellitus
- Purity(%)
- >=99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Powder
- Loss on Drying
- 5.0%
- Particle Size
- Typically 90% <10 microns
- Solubility
- Freely soluble in water, practically insoluble in ether and chloroform
- Bacterial Endotoxins
- <0.5 IU/mg
- Shelf Life
- 2 years
- Polymorphism
- No polymorphic forms detected
- Specific Rotation
- +162 to +172 (c=1, water)
- Microbial Limit
- <100 cfu/g
- Heavy Metal (%)
- 0.001%
- Poisonous
- Non-poisonous in therapeutic dose
- Related Substances
- Total impurities 1.0%
- Packaging
- Double polyethylene bags within fiber drum
- Storage
- Store at 2-8C, well sealed, protected from light
- Taste
- Slightly sweet
- Identification Method
- HPLC/IR spectroscopy
- Color
- White to off-white
- Residual Solvents
- Complies with ICH guidelines
- Ph Level
- Neutral pH (5.5 - 7) in solution
- Smell
- Odorless
About Acarbose API
Why Choose Acarbose API: Unmatched Advantages for Diabetes Care
Acarbose API offers outstanding competitive advantages, being a highly potent, pharmaceutical-grade agent specifically used to manage type 2 diabetes mellitus. Its application method involves oral administration in formulated medications, effectively regulating blood glucose levels. With a splendiferous reputation for purity, stability, and negligible total impurities, it ensures exceptional patient safety. The extraordinary solubility and finely controlled particle size enhance its therapeutic performance, making it a favored choice for healthcare providers and manufacturers seeking reliable antidiabetic solutions.
Premium Packaging, Quick Dispatch, and Quality Assurance for Acarbose
Our Acarbose API is meticulously packaged in double polyethylene bags within sturdy fiber drums, safeguarding its quality during transit. Shipped goods are dispatched promptly, ensuring minimal delivery times to meet global demands. Each batch is accompanied by comprehensive certifications, affirming its compliance with regulatory standards and sale price valuation. This rigorous approach ensures the highest industry benchmarks in purity and safety, making our Acarbose API highly valued in international markets.
FAQ's of Acarbose API:
Q: How is Acarbose API typically administered in medical applications?
A: Acarbose API is formulated into oral medications, usually in tablet form, for the management of type 2 diabetes mellitus. It is designed to be taken with the first bite of a meal to maximize its therapeutic effect on blood glucose regulation.Q: What makes Acarbose API a favorable option for diabetes treatment?
A: Acarbose API is renowned for its extraordinary purity (99%) and stringent quality controls. Its unique mechanism helps control post-meal blood sugar spikes, offering a non-poisonous, stable, and efficacious solution for type 2 diabetes care.Q: Where should Acarbose API be stored to ensure optimal stability?
A: Acarbose API should be stored at 2-8C, well sealed, and protected from light to preserve its splendiferous potency and extend its two-year shelf life.Q: What are the packaging and delivery standards for Acarbose API?
A: Acarbose API is securely packaged in double polyethylene bags within fiber drums. Shipped goods are delivered promptly, with each batch accompanied by quality certifications, ensuring a favorable sale price and valuation in accordance with international regulations.Q: What certifications and analyses are provided with Acarbose API shipments?
A: Each shipment comes with complete certification, including analysis by HPLC/IR spectroscopy, impurity profiling, microbial limit testing, and confirmation of compliance with ICH guidelines. This documentation assures that the product meets pharmaceutical-grade standards globally.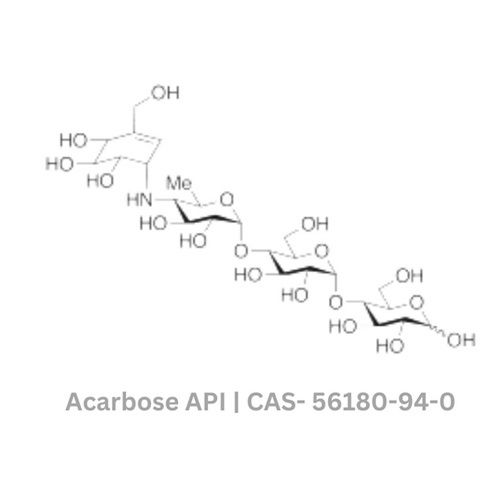

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
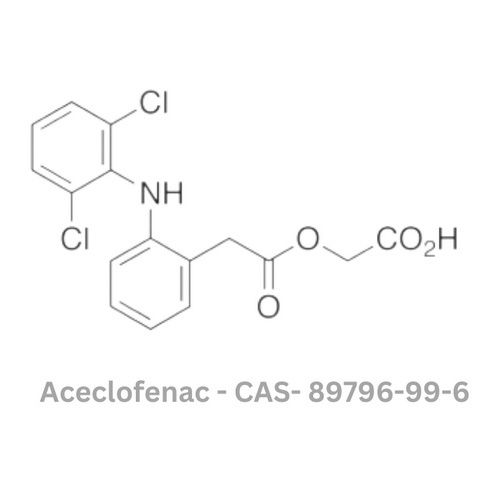
Aceclofenac API
Minimum Order Quantity : 100 Kilograms
Grade : Other, Pharmaceutical Grade
Physical Form : Powder
Storage : Other, Store in cool, dry place, protected from light
Type : Other, API (Active Pharmaceutical Ingredient)
Solubility : Practically insoluble in water, freely soluble in acetone and dimethylformamide
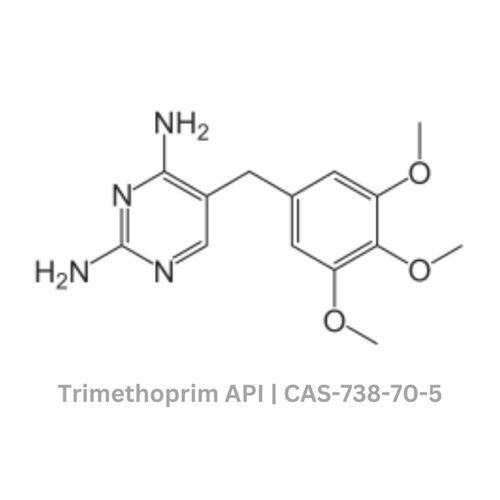
Trimethoprim API
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Storage : Other, Store in tightly closed containers, protected from light and moisture
Type : Other, Active Pharmaceutical Ingredient
Solubility : Slightly soluble in water, soluble in DMSO and ethanol
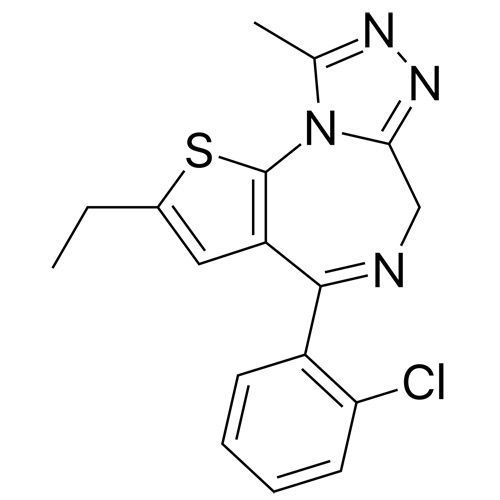
Etizo Lam API Powder
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Physical Form : Powder
Storage : Room Temperature
Type : Other, Etizo Lam API Powder
Solubility : Yes
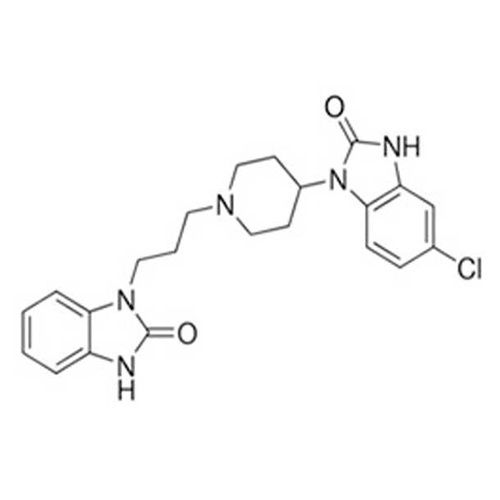
Domperidone API
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Physical Form : Powder
Storage : Room Temperature
Type : Other, Domperidone API
Solubility : Yes


 Send Inquiry
Send Inquiry