Trimethoprim API
Trimethoprim API Specification
- Smell
- Odorless
- Ph Level
- Neutral - depends on specific batch analysis
- Solubility
- Slightly soluble in water, soluble in DMSO and ethanol
- Structural Formula
- C14H18N4O3
- Color
- White to off-white
- Molecular Formula
- C14H18N4O3
- Shelf Life
- 24 Months
- Particle Size
- Specified on COA, typically micronized (e.g., D90 < 10 m)
- Molecular Weight
- 290.32 g/mol
- Taste
- Bitter
- Boiling point
- Not applicable (decomposes before boiling)
- EINECS No
- 212-019-0
- Poisonous
- No, utilized medicinally but can be toxic in overdose
- Loss on Drying
- Not more than 0.5%
- Heavy Metal (%)
- Not more than 0.001%
- HS Code
- 29420090
- Melting Point
- 199-203C
- Storage
- Store in tightly closed containers, protected from light and moisture
- Medicine Name
- Trimethoprim API
- Chemical Name
- 2,4-Diamino-5-(3,4,5-trimethoxybenzyl)pyrimidine
- CAS No
- 738-70-5
- Type
- Active Pharmaceutical Ingredient
- Grade
- Pharmaceutical Grade
- Usage
- Used in the treatment of bacterial infections, mainly urinary tract infections
- Purity(%)
- Not less than 99.0%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Polymorphism
- Single crystalline form
- Chloride
- Not more than 0.05%
- Bulk Density
- 0.30 - 0.65 g/ml
- Manufacturing Process
- Synthetic
- Identification
- By HPLC and IR Spectroscopy
- Sulphate
- Not more than 0.05%
- Residual Solvents
- Complies with ICH guidelines
- Iron
- Not more than 0.002%
- Assay
- 99.0%-101.0% (on dried basis)
- Packaging
- HDPE drums or double polyethylene bags
- Particle Size Distribution
- Available as per requirement (micronized or non-micronized)
- Microbial Limit
- Complies with pharmacopoeial standards
- Related Substances
- Not more than 0.5%
- Water Content (Karl Fischer)
- Not more than 0.5%
Trimethoprim API Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Trimethoprim API
Trimethoprim API: Extra Features & Broad Application Spectrum
Trimethoprim API stands out with its exceptional purity, single crystalline polymorph, and customizable micronized particle sizes. Its application extends across formulations for urinary tract infections, a wide range of bacterial infection therapies, and pharmaceutical research. Produced using a synthetic process, it is suitable for modern medicine manufacturing plants and research facilities. Its compliance with ICH and pharmacopoeial norms and flexible packaging options enhance its versatility for manufacturers seeking superior-grade ingredients.
Trimethoprim API: Payment, Certifications, and Delivery Insights
We guarantee secure arrival of Trimethoprim API through reliable goods transport partners. Sale price is structured competitively to ensure affordability for bulk orders. Our product holds necessary certifications, ensuring quality and regulatory compliance in pharmaceutical markets. The main domestic market covers pan-India, allowing swift supply to pharmaceutical manufacturers and authorized buyers. Detailed payment terms are provided at enquiry, supporting transparency and customer confidence. All shipments are traceable for peace of mind.
FAQ's of Trimethoprim API:
Q: How is Trimethoprim API identified and what analytical methods are employed?
A: Trimethoprim API is identified using high-performance liquid chromatography (HPLC) and IR spectroscopy, ensuring accurate verification of its purity and structural attributes before release.Q: What are the primary usage areas of Trimethoprim API?
A: Trimethoprim API is mainly used in the formulation of medicines targeting bacterial infections, especially urinary tract infections, and plays a key role in pharmaceutical and clinical research applications.Q: Where can buyers find details about the purity and particle size of this API?
A: All critical specifications, including purity and particle size distribution, are provided on the Certificate of Analysis (COA) accompanying each shipment, tailored to client requirements.Q: What certifications and quality standards does Trimethoprim API comply with?
A: Trimethoprim API complies with Indian and international pharmacopoeial standards, and all batches are tested to meet ICH guidelines for residual solvents and microbial limits for pharmaceutical applications.Q: When is the best time to place an order to ensure prompt arrival and best price?
A: Placing orders during scheduled production cycles or bulk procurement periods ensures timely goods transport, competitive sale prices, and assures the earliest possible delivery windows.Q: How should Trimethoprim API be stored to maintain its quality and shelf life?
A: For optimal stability, Trimethoprim API should be stored in tightly sealed containers, protected from moisture and direct light, preserving its 24-month shelf life.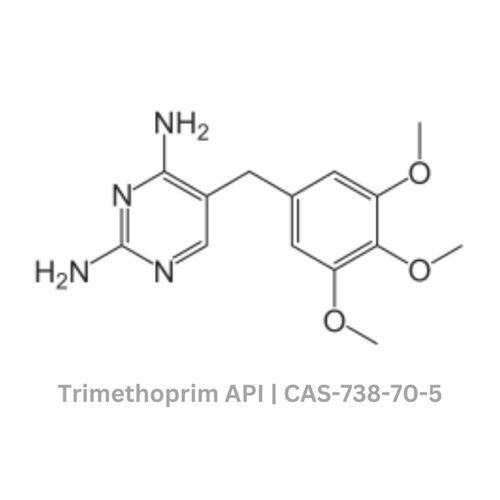

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
Rivaroxaban API
Minimum Order Quantity : 100 Pieces
Grade : Other, Pharmaceutical Grade
Storage : Other, Store in a cool, dry, wellventilated area, away from direct sunlight
Physical Form : Solid
Type : Other, Active Pharmaceutical Ingredient (API)
Purity(%) : >=99%
Hydrochloride Monohydrate
Grade : Other, Pharmaceutical
Storage : Other, Store in cool, dry place
Physical Form : Solid
Type : Other, Active Pharmaceutical Ingredient (API)
Purity(%) : 99.8%
Gliclazide API
Grade : Other, Pharmaceutical Grade
Storage : Other, Store in a cool, dry place, protected from light
Physical Form : Solid
Type : Other, Active Pharmaceutical Ingredient
Purity(%) : 99%
Itraconazole API CAS No: 84625-61-6
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Storage : Room Temperature
Physical Form : Powder
Type : Other, Itraconazole API


 Send Inquiry
Send Inquiry