Gliclazide API
Gliclazide API Specification
- Poisonous
- Non-poisonous under recommended usage
- Structural Formula
- C15H21N3O3S
- Solubility
- Freely soluble in dimethylformamide, sparingly soluble in methanol, practically insoluble in water
- Loss on Drying
- Not more than 0.5%
- EINECS No
- 244-277-4
- Melting Point
- 168-170C
- HS Code
- 29420090
- Particle Size
- D90 <10 microns (customizable)
- Taste
- Bitter
- Molecular Formula
- C15H21N3O3S
- Heavy Metal (%)
- Not more than 0.001%
- Color
- White
- Smell
- Odorless
- Shelf Life
- 36 months
- Molecular Weight
- 323.41 g/mol
- Storage
- Store in a cool, dry place, protected from light
- Medicine Name
- Gliclazide API
- Chemical Name
- Gliclazide
- CAS No
- 21187-98-4
- Type
- Active Pharmaceutical Ingredient
- Grade
- Pharmaceutical Grade
- Usage
- Used as an oral hypoglycemic agent in the treatment of type 2 diabetes mellitus
- Purity(%)
- 99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Identification
- Complies with standard USP/EP identification tests
- Packaging
- Double polyethylene bags with HDPE drum
- Regulatory Compliance
- US FDA, EDQM, WHO-GMP
- Residue on Ignition
- Not more than 0.1%
- Microbial Limit
- Complies with pharmacopeial standards
- Manufacturing Process
- Synthetic
- Retest Period
- 36 months from date of manufacture
- Other Names
- Diamicron
- Stability
- Stable under normal conditions
- Related Substances
- Not more than 1.0%
- Assay (by HPLC)
- 98.0% 101.0%
- Polymorphic Form
- Form I (standard)
Gliclazide API Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Gliclazide API
Exceptional Application Flexibility and Plant Integration
Gliclazide API is extensively used in pharmaceutical manufacturing plants for formulating oral hypoglycemic agents targeting type 2 diabetes mellitus. Its customizable particle size (D90 <10 microns) enhances plant adaptability, supporting various dosage forms. Features such as high assay purity, robust polymorphic stability (Form I), and negligible residue on ignition confirm its suitability for both large and small-scale production facilities, ensuring optimal effectiveness wherever applied within global pharma plants.
Certified Exports, Strategic Markets, and Protective Packaging
Our Gliclazide API is US FDA, EDQM, and WHO-GMP certified, making it eligible for both domestic and international markets. With established freight channels, we serve the main domestic markets across India and export to numerous global destinations efficiently. The product is securely packaged in double-layered polyethylene bags within HDPE drums, minimizing damage during transit and shipment. Charges are transparently communicated, ensuring worry-free, compliant delivery from manufacturer to end-user worldwide.
FAQ's of Gliclazide API:
Q: How is the quality of Gliclazide API ensured during manufacturing and packaging?
A: The Gliclazide API undergoes rigorous quality checks with compliance to USP/EP identification tests, and is packaged in double polyethylene bags within HDPE drums to protect its stability and purity during transport and storage.Q: What makes this Gliclazide API meritorious for pharmaceutical manufacturers?
A: Its matchless purity of 99%, stability under normal conditions, and comprehensive regulatory compliance (US FDA, EDQM, WHO-GMP) make this API especially suitable for formulators demanding high-quality active ingredients for their products.Q: Where can Gliclazide API be applied within pharmaceutical production environments?
A: It is primarily used in the production of oral hypoglycemic agents in plants that manufacture medicines for type 2 diabetes mellitus, with versatile particle size options to integrate seamlessly into various drug formulations.Q: When should the Gliclazide API be retested for quality assurance?
A: The retest period is up to 36 months from the date of manufacture, as long as the API is stored under recommended conditions (cool, dry place, protected from light).Q: How does the freight and packaging process preserve product quality during export?
A: Carefully designed packaging-double-layered polyethylene bags inside HDPE drums-coupled with efficient freight logistics, ensures the product reaches both domestic and international clients intact, maintaining its quality through the supply chain.Q: What benefits does the customizable particle size of Gliclazide API offer?
A: Customizable particle size (D90 <10 microns) allows formulators to tailor the API for optimal dissolution and bioavailability in specific dosage forms, maximizing therapeutic effectiveness and production efficiency.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
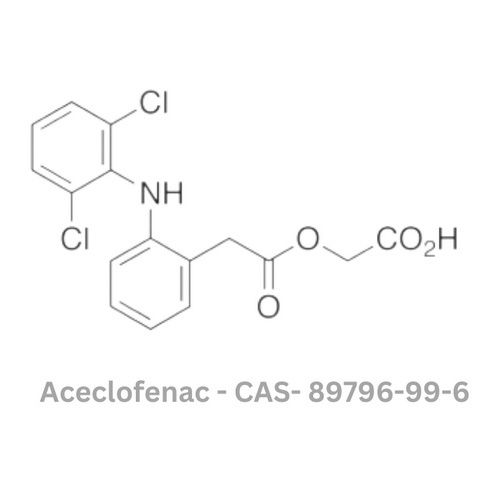
Aceclofenac API
Minimum Order Quantity : 100 Kilograms
Physical Form : Powder
Grade : Other, Pharmaceutical Grade
Smell : Other, Odorless
Taste : Other , Slightly bitter
Type : Other, API (Active Pharmaceutical Ingredient)
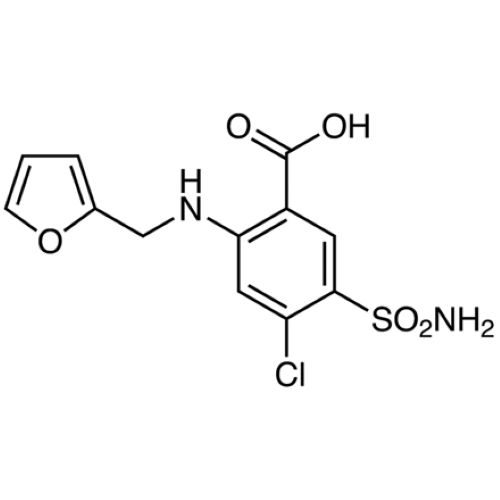
Furosemide IP/BP/EP/USP
Minimum Order Quantity : 100 Kilograms
Physical Form : Powder
Grade : Medicine Grade
Smell : No Smell
Taste : Odorless
Type : Pharmaceutical Intermediates
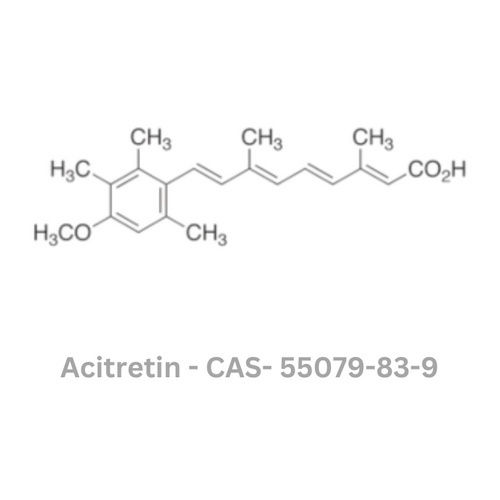
Acitretin API
Minimum Order Quantity : 100 Kilograms
Physical Form : Powder
Grade : Industrial Grade
Smell : Sharp
Taste : Odorless
Type : Other, Acitretin API
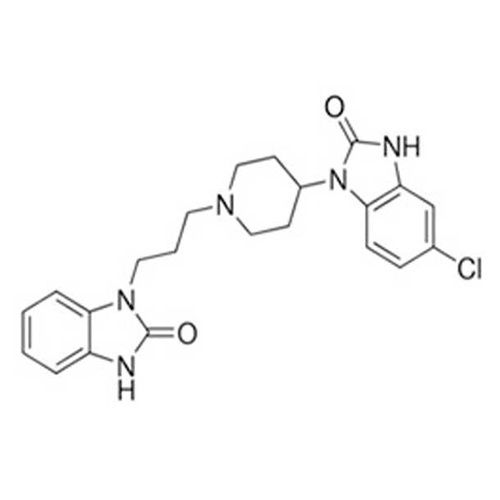
Domperidone API
Minimum Order Quantity : 100 Kilograms
Physical Form : Powder
Grade : Industrial Grade
Smell : Stimulus
Taste : Bitter
Type : Other, Domperidone API


 Send Inquiry
Send Inquiry