Furosemide IP/BP/EP/USP
Price 2300.0 INR/ Kilograms
Furosemide IP/BP/EP/USP Specification
- Melting Point
- 206-220
- EINECS No
- 200-203-6
- Molecular Formula
- C12-H11-ClN2-O5S
- Boiling point
- C
- HS Code
- 29359090
- Smell
- No Smell
- Taste
- Odorless
- Molecular Weight
- 330.74 GSM (gm/2)
- Chemical Name
- Furosemide
- CAS No
- 54-31-9
- Type
- Pharmaceutical Intermediates
- Grade
- Medicine Grade
- Usage
- Furosemide API is widely utilized in the pharmaceutical industry to produce tablets and injection formulations to reduce swelling and fluid overload in congestive heart failure and renal conditions.
- Purity(%)
- 99.99
- Appearance
- Almost white to yellow crystalline powder, odorless, and almost tasteless.
- Physical Form
- Powder
Furosemide IP/BP/EP/USP Trade Information
- Minimum Order Quantity
- 100 Kilograms
- Supply Ability
- 1000 Kilograms Per Month
- Delivery Time
- 15 Days
About Furosemide IP/BP/EP/USP
- Appearance: Almost white to yellow crystalline powder, odorless, and almost tasteless.
- Solubility: Soluble in acetone, sparingly soluble in ethanol, and insoluble in water.
- Mechanism of Action: Inhibits the NKCC2 (Na-K-Cl) cotransporter in the thick ascending limb of the loop of Henle, resulting in rapid diuresis.
- Grade: Typically supplied under Good Manufacturing Practice (GMP) compliant conditions for pharmaceutical formulation.
Furosemide API is widely utilized in the pharmaceutical industry to produce tablets and injection formulations to reduce swelling and fluid overload in congestive heart failure and renal conditions.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
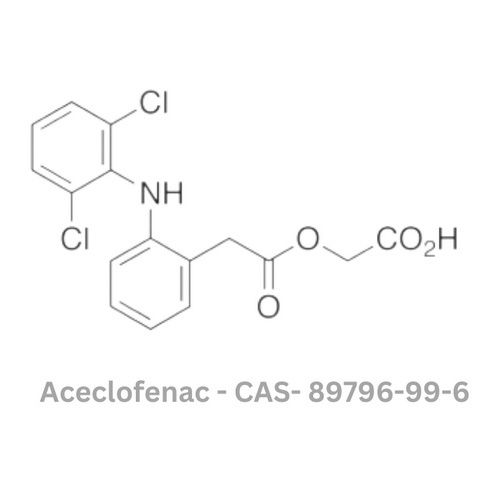
Aceclofenac API
Minimum Order Quantity : 100 Kilograms
Smell : Other, Odorless
Solubility : Practically insoluble in water, freely soluble in acetone and dimethylformamide
Type : Other, API (Active Pharmaceutical Ingredient)
Physical Form : Powder
Storage : Other, Store in cool, dry place, protected from light
Ursodeoxycholic Acid
Minimum Order Quantity : 100 Kilograms
Smell : Other, Odorless
Solubility : Slightly soluble in water, freely soluble in ethanol and acetone
Type : Other, Active Pharmaceutical Ingredient
Physical Form : Solid
Storage : Other, Store in a cool, dry place in tightly closed containers
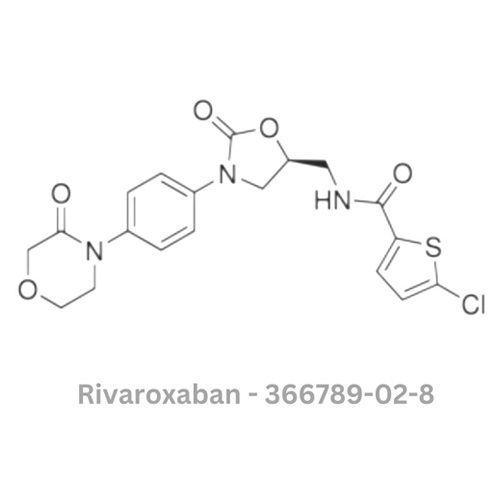
Rivaroxaban API
Minimum Order Quantity : 100 Pieces
Smell : Other, Odourless
Solubility : Slightly soluble in water; soluble in organic solvents
Type : Other, Active Pharmaceutical Ingredient (API)
Physical Form : Solid
Storage : Other, Store in a cool, dry, wellventilated area, away from direct sunlight
Salbutamol Sulphate
Smell : Other, Odorless
Solubility : Freely soluble in water
Type : Other, Active Pharmaceutical Ingredient
Physical Form : Powder
Storage : Other, Store in cool, dry place, protect from light


 Send Inquiry
Send Inquiry