Salbutamol Sulphate
Salbutamol Sulphate Specification
- Molecular Formula
- C13H21NO3S
- Solubility
- Freely soluble in water
- EINECS No
- 256-916-8
- Structural Formula
- Available on Request
- Shelf Life
- 5 years
- Melting Point
- 165-171C
- Loss on Drying
- <0.5%
- Color
- White
- Storage
- Store in cool, dry place, protect from light
- Taste
- Characteristic
- HS Code
- 29420090
- Heavy Metal (%)
- <0.001%
- Smell
- Odorless
- Poisonous
- NO
- Ph Level
- 4.0-5.5
- Molecular Weight
- 287.38 g/mol
- Particle Size
- Micronized
- Medicine Name
- Salbutamol Sulphate
- Chemical Name
- Salbutamol Sulphate
- CAS No
- 51022-70-9
- Type
- Active Pharmaceutical Ingredient
- Grade
- Pharma Grade
- Usage
- Anti-asthmatic; Bronchodilator
- Purity(%)
- 99% min
- Appearance
- White to off-white powder
- Physical Form
- Powder
- Residue on Ignition
- <0.2%
- Optical Rotation
- +6.0° to +8.0° (C=1, H2O)
- Packing
- HDPE Drum / Double PE Bags
- Manufacturing Process
- Synthetic
- Related Substances
- Total impurities <1.0%
- Identification
- IR/UV conforms
- Assay (by HPLC)
- 98.0%-102.0%
- Microbial Limit
- Standard pharmacopeia limits
Salbutamol Sulphate Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Salbutamol Sulphate
- Generic Name : Salbutamol Sulphate API
- CAS Number : 51022-70-9
- Grade : IP / BP / USP
- Packing Type : Drum
- Therapeutic use : Bronchospasm / Airway secretion clearance therapy
- Product MOQ : 25 Kg
Premium Pharmaceutical Quality
Salbutamol Sulphate meets stringent pharmaceutical grade standards, validated by comprehensive testing such as assay by HPLC (98.0%-102.0%) and impurity controls (<1.0%). Its safety profile is reinforced by conformity with microbial limits, making it a reliable choice for medicinal formulations.
Optimal Physical Properties
The API presents as a micronized white to off-white powder, ensuring consistent particle size and ease of formulation. With a Ph level of 4.0-5.5, and a melting point range of 165-171C, Salbutamol Sulphate is both stable and practical for various pharmaceutical processes.
Secure & Lasting Packaging
Salbutamol Sulphate is packed in HDPE drums and double PE bags, providing robust protection during storage and transportation. This packaging approach minimizes contamination risks and preserves the ingredient's potency for up to 5 years when stored properly.
FAQ's of Salbutamol Sulphate:
Q: How is Salbutamol Sulphate typically used in pharmaceutical applications?
A: Salbutamol Sulphate is primarily formulated as a bronchodilator and anti-asthmatic agent, serving in medications that help alleviate respiratory distress associated with asthma and other pulmonary conditions.Q: What benefits does its high purity and micronized form offer?
A: A purity of at least 99% and micronized particle size ensure superior efficacy, precise dosage, and enhanced solubility, contributing to improved patient outcomes in respiratory therapies.Q: What is the recommended storage condition for Salbutamol Sulphate?
A: It should be kept in a cool, dry place, protected from light, to maintain stability and potency for up to five years from the date of manufacture.Q: Where can Salbutamol Sulphate be sourced from?
A: This ingredient is manufactured and exported by reputable suppliers in India, who adhere to global pharmaceutical standards in production and packaging.Q: What process is used to manufacture Salbutamol Sulphate?
A: Salbutamol Sulphate is produced using a synthetic process that ensures consistent quality, controlled impurity levels, and reliable batch reproducibility.Q: Are there safety considerations regarding toxicity?
A: Salbutamol Sulphate is non-poisonous when used as intended in pharmaceutical applications and contains minimal heavy metals (<0.001%), adhering to international safety standards.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
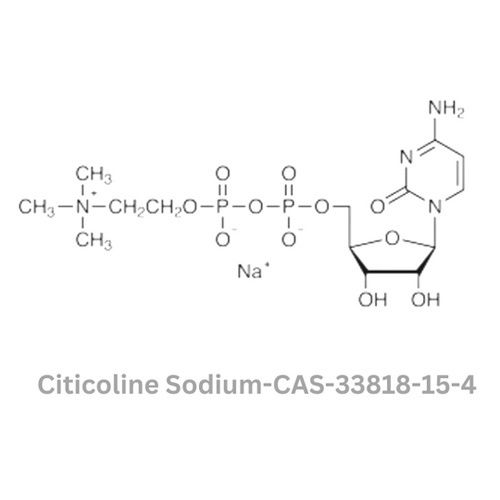
Citicoline Sodium
Minimum Order Quantity : 100 Kilograms
Grade : Other, Pharmaceutical Grade
Physical Form : Powder
Storage : Other, Store in a cool, dry place away from light and moisture
Smell : Other, Odorless
Color : Other, White to offwhite
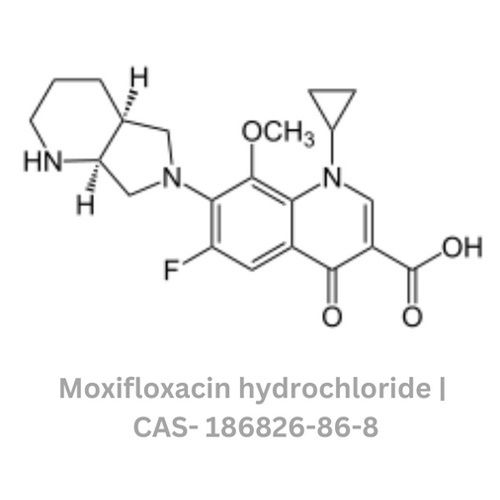
Moxifloxacin hydrochloride
Minimum Order Quantity : 100 Kilograms
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Storage : Other, Store in a tightly closed container at room temperature, away from excess heat and moisture
Smell : Other, Odorless
Color : Other, White to pale yellow
Ursodeoxycholic Acid
Minimum Order Quantity : 100 Kilograms
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Storage : Other, Store in a cool, dry place in tightly closed containers
Smell : Other, Odorless
Color : White
Hydroxychloroquine Sulfate API Powder
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Physical Form : Solid
Storage : Room Temperature
Smell : Stimulus
Color : White


 Send Inquiry
Send Inquiry