Citicoline Sodium
Citicoline Sodium Specification
- Boiling point
- Not available (decomposes before boiling)
- Poisonous
- Non-poisonous at therapeutic doses
- EINECS No
- 251-689-1
- Storage
- Store in a cool, dry place away from light and moisture
- Heavy Metal (%)
- Not more than 0.001%
- Solubility
- Freely soluble in water, practically insoluble in ethanol and methylene chloride
- Particle Size
- NMT 150 microns
- Molecular Weight
- 510.32 g/mol
- HS Code
- 29241990
- Smell
- Odorless
- Melting Point
- Not less than 200C (decomposes)
- Molecular Formula
- C14H25N4NaO11P2
- Color
- White to off-white
- Taste
- Slight saline
- Loss on Drying
- Not more than 6.0%
- Structural Formula
- C14H25N4NaO11P2
- Ph Level
- 6.0 to 7.5 (in 2% solution)
- Shelf Life
- 36 months
- Medicine Name
- Citicoline Sodium
- Chemical Name
- Cytidine-5-diphosphocholine sodium salt
- CAS No
- 33818-15-4
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Used in the treatment of stroke, cognitive impairment, and neurological disorders
- Purity(%)
- 98.0% Min
- Appearance
- White to off-white crystalline powder
- Physical Form
- Powder
- Packaging
- Double polyethylene bags in fiber drum
- Identification
- Complies with standard tests (IR, HPLC, UV)
- Assay (on dried basis)
- 98-102%
- Bacterial Endotoxins
- <0.25 EU/mg
- Related Substances
- Total impurities not more than 2.0%
- Microbial Limit
- Total aerobic microbial count NMT 1000 CFU/g, Total Yeast and Molds NMT 100 CFU/g
- Residual Solvent
- Within ICH limits
- Application
- Pharmaceutical, Nutraceutical formulations
Citicoline Sodium Trade Information
- Minimum Order Quantity
- 100 Kilograms
- Supply Ability
- 1000 Kilograms Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Citicoline Sodium
Citicoline Sodium: Plant Application & Competitive Edge
Citicoline Sodium is expertly formulated to deliver potent neuroprotection, making it ideal for inclusion in pharmaceutical and nutraceutical manufacturing plants. Its competitive advantages include high assay compliance, minimal impurities, and robust microbial limits. Application is versatile-simply dissolve the powder in aqueous solutions to prepare formulations for cognitive enhancement or stroke recovery supplements. This API's reliable solubility, stringent quality control, and stability offer manufacturing plants a robust ingredient to ensure the merit of every batch produced.
Domestic Market, Packaging & Certifications: An Investment in Quality
Exchange robust value when sourcing Citicoline Sodium, supplied across India's main pharmaceutical and nutraceutical markets. Secure double polyethylene bags in sturdy fiber drums maintain product efficacy and meet international export standards. Obtain a formal quotation to plan your outlay, knowing each package is supported by thorough quality certifications and regulatory compliance. Rely on our documentation for seamless import/export processes and confident usage in high-standard medicinal product manufacturing environments.
FAQ's of Citicoline Sodium:
Q: How should Citicoline Sodium be stored to ensure maximum shelf life?
A: Citicoline Sodium should be stored in a cool, dry place away from light and moisture. When kept under recommended conditions, it maintains its quality for up to 36 months from the date of manufacture.Q: What are the primary applications of Citicoline Sodium in the pharmaceutical industry?
A: Citicoline Sodium is primarily used as an Active Pharmaceutical Ingredient in medicines treating stroke, cognitive impairment, and neurological disorders. It is also widely utilized in nutraceutical formulations aiming to enhance cognitive function.Q: When can a buyer expect product delivery after placing an order?
A: Delivery timelines depend on current inventory and destination. However, orders are processed promptly upon confirmation, and shipment details-including tracking-are provided for transparency.Q: Where is Citicoline Sodium manufactured and exported from?
A: Our high-purity Citicoline Sodium is manufactured and supplied from India, and we handle both domestic supply and international export to meet global demand.Q: What purification and quality identification processes are followed?
A: Each batch undergoes comprehensive identification tests using IR, HPLC, and UV methods. Rigorous assays and impurity analysis are performed to ensure compliance with pharmaceutical-grade standards, and all parameters are aligned with ICH guidelines and microbial safety limits.Q: How is Citicoline Sodium typically added to product formulations?
A: Citicoline Sodium is commonly dissolved in water-based solutions for ease of integration into pharmaceutical or nutraceutical product manufacturing, thanks to its excellent water solubility and precise particle size control.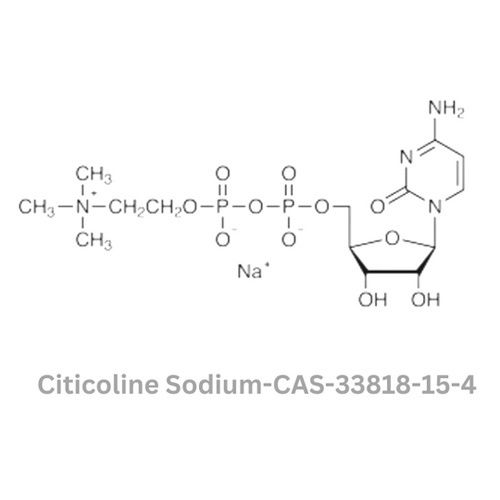

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
Levocetirizine DI HCL API
Minimum Order Quantity : 100 Kilograms
Type : Other, Levocetirizine DI HCL API
Solubility : Freely soluble in water, practically insoluble in acetone and in methylene chloride
Physical Form : Solid
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade
Bupivacaine Hydrochloride
Type : Other, Active Pharmaceutical Ingredient (API)
Solubility : Freely soluble in water and alcohol
Physical Form : Solid
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade
Dexchlorpheniramine Maleate
Type : Other, API (Active Pharmaceutical Ingredient)
Solubility : Freely soluble in water and alcohol
Physical Form : Powder
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade
Granisetron Hydrochloride
Type : Other, Active Pharmaceutical Ingredient (API)
Solubility : Freely soluble in water, soluble in methanol, slightly soluble in ethanol.
Physical Form : Solid
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade


 Send Inquiry
Send Inquiry