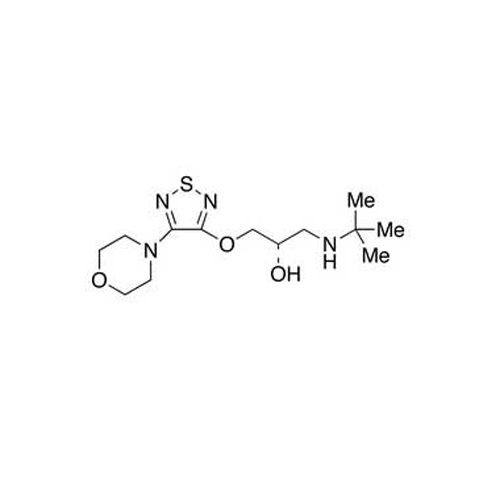
Granisetron Hydrochloride
Granisetron Hydrochloride Specification
- Storage
- Store in a cool, dry place, protected from light
- Particle Size
- As per customer requirement (typically micronized)
- Taste
- Bitter
- Color
- White to off-white
- Molecular Formula
- C18H25ClN4O
- Shelf Life
- 36 months
- Solubility
- Freely soluble in water, soluble in methanol, slightly soluble in ethanol.
- Molecular Weight
- 348.87 g/mol
- Poisonous
- Non-poisonous at therapeutic doses
- Heavy Metal (%)
- NMT 0.001%
- Melting Point
- 218-220C
- Ph Level
- 4.0-6.0 (1% solution)
- HS Code
- 29349900
- Structural Formula
- C18H25ClN4O
- EINECS No
- 616-977-2
- Smell
- Odorless
- Loss on Drying
- NMT 0.5%
- Medicine Name
- Granisetron Hydrochloride
- Chemical Name
- Granisetron Hydrochloride
- CAS No
- 107007-99-8
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Used as an antiemetic to prevent nausea and vomiting caused by chemotherapy, radiation, or surgery
- Purity(%)
- 99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Endotoxin Level
- < 0.5 EU/mg
- Assay (on dried basis)
- 98.0%
- Identification
- By IR and HPLC
- Water Content (by Karl Fischer)
- NMT 0.5%
- Residual Solvents
- Complies with ICH guidelines
- Chloride Content
- 13.0-13.6%
- Bulk Density
- 0.30-0.50 g/cm
- Packing
- Double polyethylene bags placed in HDPE drums
- Related Substances
- NMT 0.2%
Granisetron Hydrochloride Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Granisetron Hydrochloride
Granisetron Hydrochloride: Versatile and Reliable Solution
Granisetron Hydrochloride is highly suited for use as an antiemetic, effectively preventing nausea and vomiting caused by chemotherapy, radiation, or surgery. Noted for its superior features-high purity, compliance with rigorous ICH standards, and optimal solubility-it is favored by pharmaceutical manufacturers, medical distributors, and healthcare providers alike. Its non-poisonous nature at therapeutic doses and extended 36-month shelf life make it a trusted choice for critical care preparations.
Supply Ability, Pricing, and Global Export Markets
With robust supply ability and flexible shipment arrangements, Granisetron Hydrochloride is available globally at competitive list prices. Buyers can inquire for the latest asking price based on volume and preferred payment terms, ensuring accessible transactions. As a leading exporter, manufacturer, and supplier from India, our main export markets span North America, Europe, Asia, and beyond, guaranteeing swift delivery and premier service for all pharmaceutical partners.
FAQ's of Granisetron Hydrochloride:
Q: How should Granisetron Hydrochloride be stored for maximum stability?
A: Granisetron Hydrochloride should be kept in a cool, dry place, away from direct light, to maintain its 36-month shelf life and assure consistent potency.Q: What are the main benefits of using Granisetron Hydrochloride?
A: The superior purity, compliance with ICH guidelines, and reliable antiemetic efficacy against chemotherapy, radiation, or surgery-induced nausea make it an ideal choice for pharmaceutical applications.Q: When is the optimal time to use Granisetron Hydrochloride in therapy?
A: It is typically administered prior to or immediately before exposure to chemotherapy, radiation, or surgery to effectively prevent nausea and vomiting in patients.Q: Where is Granisetron Hydrochloride primarily exported from?
A: Granisetron Hydrochloride is mainly exported from India, with reach into major global markets including North America, Europe, and Asia.Q: What processes ensure the quality of Granisetron Hydrochloride?
A: Rigorous identification (IR & HPLC), strict limits on related substances and water content, and compliance with ICH guidelines collectively maintain the product's high quality and purity.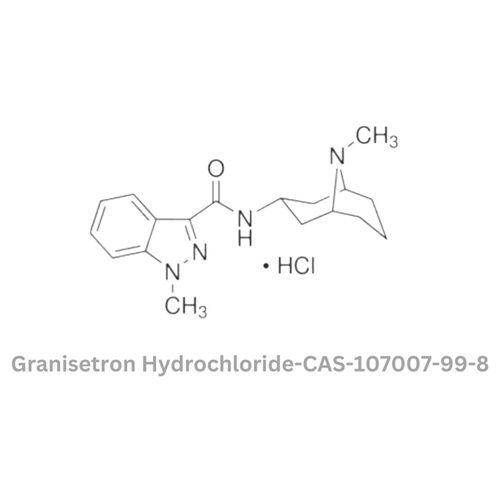

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
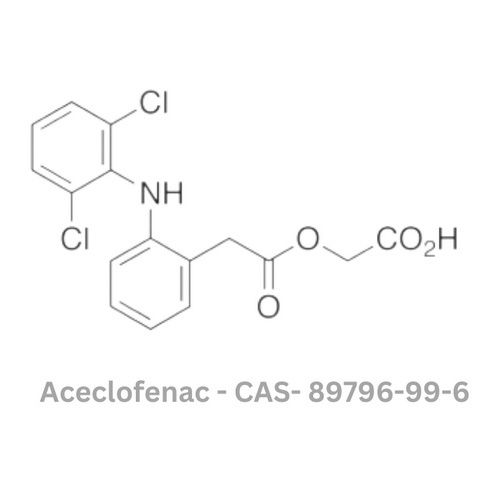
Aceclofenac API
Minimum Order Quantity : 100 Kilograms
Type : Other, API (Active Pharmaceutical Ingredient)
Grade : Other, Pharmaceutical Grade
Storage : Other, Store in cool, dry place, protected from light
Solubility : Practically insoluble in water, freely soluble in acetone and dimethylformamide
Physical Form : Powder
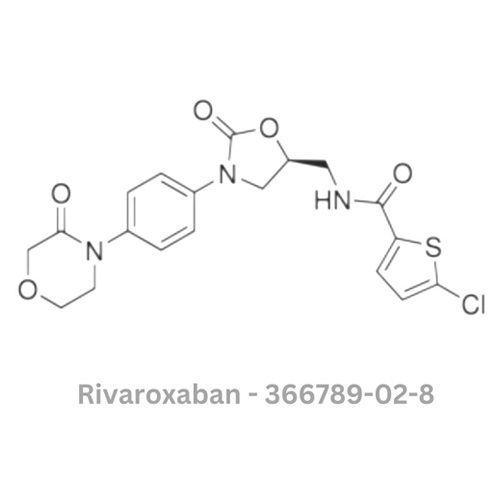
Rivaroxaban API
Minimum Order Quantity : 100 Pieces
Type : Other, Active Pharmaceutical Ingredient (API)
Grade : Other, Pharmaceutical Grade
Storage : Other, Store in a cool, dry, wellventilated area, away from direct sunlight
Solubility : Slightly soluble in water; soluble in organic solvents
Physical Form : Solid
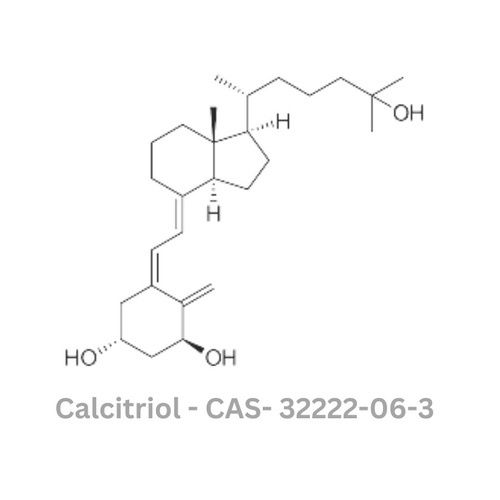
Calcitriol API
Type : Other, Active Pharmaceutical Ingredient (API)
Grade : Other, Pharmaceutical Grade
Storage : Other, Store at 28C, protected from light
Solubility : Freely soluble in organic solvents (ethanol, methanol, chloroform, ether); insoluble in water
Physical Form : Solid
Timolol Maleate API
Minimum Order Quantity : 100 Kilograms
Type : Other, Timolol Maleate API
Grade : Industrial Grade
Storage : Room Temperature
Solubility : Yes
Physical Form : Powder


 Send Inquiry
Send Inquiry