Calcitriol API
Calcitriol API Specification
- Loss on Drying
- <0.5%
- Storage
- Store at 2-8C, protected from light
- HS Code
- 29362990
- Poisonous
- Non-Poisonous at recommended dosage
- Molecular Formula
- C27H44O3
- Color
- White to off-white
- Taste
- Tasteless
- Heavy Metal (%)
- <0.001%
- Smell
- Odorless
- Boiling point
- Not applicable (decomposes before boiling)
- EINECS No
- 250-963-8
- Shelf Life
- 2 Years
- Solubility
- Freely soluble in organic solvents (ethanol, methanol, chloroform, ether); insoluble in water
- Structural Formula
- C27H44O3
- Molecular Weight
- 416.64 g/mol
- Ph Level
- Neutral
- Particle Size
- Micronized, 90% <15 m
- Melting Point
- 120-125 C
- Medicine Name
- Calcitriol
- Chemical Name
- (1,3,25-Trihydroxycholecalciferol)
- CAS No
- 32222-06-3
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Vitamin D analogue, used to treat calcium deficiency and metabolic bone disease
- Purity(%)
- >99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Microbial Limit
- Meets Pharmacopoeia standards
- Assay Method
- HPLC
- Related Substances
- Not more than 0.5%
- Identification
- Complies with standard UV and IR absorption
- Expiration Dates
- Indicated on label
- Optical Rotation
- +83° to +89° (c=1, CHCl3)
- Packaging
- Packed in light-resistant, airtight containers
- Stability
- Stable under recommended storage conditions
- Residual Solvents
- Complies with ICH guidelines
- Applications
- Pharmaceutical formulations, vitamin D preparation
Calcitriol API Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Calcitriol API
Calcitriol API: Precision Formulations for Healthcare Excellence
Calcitriol API is a pharmaceutical-grade active ingredient widely recognized for its efficacy in vitamin D preparations. Used primarily by pharmaceutical companies and compounding pharmacies, it is a crucial component for treating calcium deficiency and metabolic bone disorders. This micronized, high-purity API facilitates seamless integration into tablets, capsules, and injectable formulations, ensuring reliable performance and patient outcomes. Its application especially benefits individuals with chronic kidney disease, osteoporosis, or hypoparathyroidism, who depend on precise vitamin D analogues for therapeutic success.
Sample Policy, Dispatch, and Global Supply Options
We offer convenient sampling of Calcitriol API to support client valuation of our rarefied product quality. Quick dispatch is guaranteed upon agreement, leveraging robust supply chains and instant checkout protocols. Our FOB port, located in India, serves clients worldwide, with a distinguished supply ability fulfilling both bulk and small-scale requirements. Transparent transaction processes, competitive offers, and responsive logistics combine to ensure your procurement is both cost-effective and timely.
FAQ's of Calcitriol API:
Q: How should Calcitriol API be stored for optimal stability?
A: Calcitriol API should be stored at 2-8C, protected from light, in its original, airtight, light-resistant containers to maintain stability and potency for up to 2 years.Q: What pharmaceutical applications does Calcitriol API serve?
A: Calcitriol API is primarily used in vitamin D preparations for treating calcium deficiency, metabolic bone disease, and related disorders, playing a vital role in pharmaceutical formulations.Q: When is the expiration date indicated for shipped batches?
A: All Calcitriol API shipments have clearly marked expiration dates on their labels, ensuring clients receive products with validated shelf life and compliance.Q: Where is your FOB port for international orders?
A: Our FOB port for all international Calcitriol API orders is located in India, facilitating efficient global export and import operations.Q: What assay and purity specifications does your Calcitriol API meet?
A: Our Calcitriol API exceeds 99% purity, is analyzed by HPLC, and complies with stringent standards for related substances, residual solvents, and microbial limits.Q: How quickly can Calcitriol API samples be dispatched after a request?
A: Sample requests are addressed promptly, with quick dispatch scheduled as soon as valuations are confirmed and relevant compliance documents are arranged.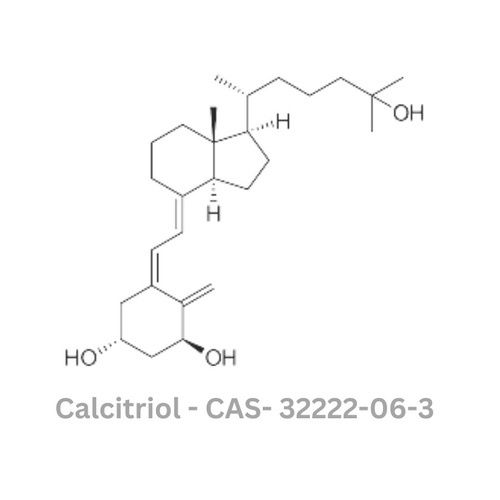

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
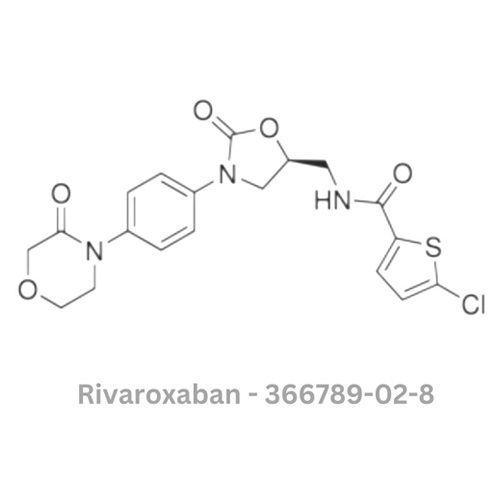
Rivaroxaban API
Minimum Order Quantity : 100 Pieces
Color : Other, White to offwhite
Type : Other, Active Pharmaceutical Ingredient (API)
Physical Form : Solid
Grade : Other, Pharmaceutical Grade
Smell : Other, Odourless
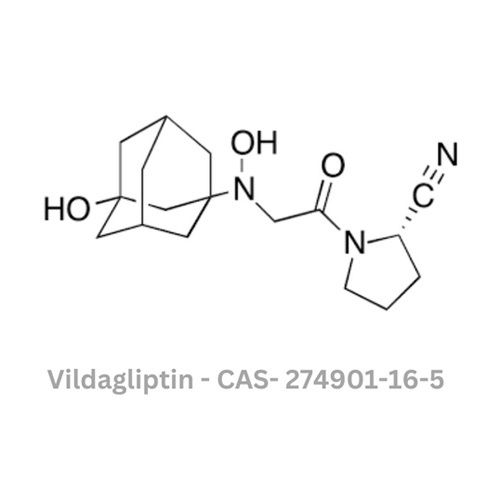
Vildagliptin API
Color : Other, White to offwhite
Type : Other, Active Pharmaceutical Ingredient (API)
Physical Form : Solid
Grade : Other, Pharmaceutical Grade
Smell : Other, Odorless
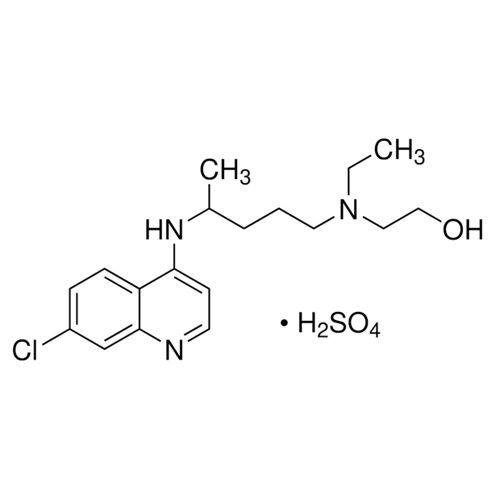
Hydroxychloroquine Sulfate API Powder
Minimum Order Quantity : 100 Kilograms
Color : White
Type : Other, Hydroxychloroquine Sulfate API Powder
Physical Form : Solid
Grade : Industrial Grade
Smell : Stimulus
Hyoscine Butylbromide API
Minimum Order Quantity : 100 Kilograms
Color : White
Type : Other, Hyoscine Butylbromide API
Physical Form : Powder
Grade : Industrial Grade
Smell : Stimulus


 Send Inquiry
Send Inquiry