Vildagliptin API
Vildagliptin API Specification
- Poisonous
- NO
- Structural Formula
- Available on request
- Solubility
- Freely soluble in methanol, slightly soluble in water
- Molecular Formula
- C17H25N3O2
- Heavy Metal (%)
- 0.001% (10 ppm)
- Molecular Weight
- 303.40 g/mol
- HS Code
- 29420090
- Boiling point
- Not applicable (decomposes)
- Storage
- Store in a cool, dry place, protected from light
- Melting Point
- 209-214C
- Taste
- Bitter
- Shelf Life
- 2 years from date of manufacture
- Particle Size
- D90 < 150 microns
- Color
- White to off-white
- Smell
- Odorless
- Loss on Drying
- 0.5%
- Medicine Name
- Vildagliptin
- Chemical Name
- (S)-1-[N-(3-Hydroxy-1-adamantyl)glycyl]pyrrolidine-2-carbonitrile
- CAS No
- 274901-16-5
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Antidiabetic agent, used in the treatment of type 2 diabetes mellitus
- Purity(%)
- >= 99%
- Appearance
- White to off-white powder
- Physical Form
- Solid
- Residual Solvents
- Within ICH limits
- Microbial Limits
- Total aerobic microbial count: < 1000 CFU/g, Yeast & mold: < 100 CFU/g
- Stability Data
- Available on request
- Identification
- By HPLC and IR spectroscopy
- Packaging
- HDPE Drums with double polyethylene bags, 1 kg/5 kg/10 kg packing or as required
- Impurities
- Total impurities 0.2%
- Regulatory Support
- DMF/CTD available on request
- End Use
- As an antidiabetic agent for pharmaceutical formulations
- Manufacturing Standard
- GMP compliant facility
- Specific Optical Rotation
- + 44 to + 48 (c=1 in methanol)
- Certificate of Analysis (COA)
- Available with each batch
Vildagliptin API Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Vildagliptin API
Vildagliptin API: Powerful Antidiabetic Excellence
Vildagliptin API is used for the effective management of type 2 diabetes mellitus, delivering pronounced glycemic control in pharmaceutical formulations. Its competitive advantages include breathtaking purity, stringent impurity limits, and compliance with global standards. This first-rate API is trusted by major health institutions, leading medicine manufacturers, and innovative research organizations worldwide, who demand high efficacy for patient care. Choose Vildagliptin for grandiose reliability and performance in diabetic therapeutics.
Export Markets, Shipping, and Packaging Details for Vildagliptin API
Our Vildagliptin API is shipped to prime export markets including North America, Europe, Southeast Asia, and Africa, ensuring broad international reach. For your assurance, samples are available free of charge and processed promptly for evaluation. Each batch is securely packaged in HDPE drums with double polyethylene bags (1/5/10 kg or as required), ensuring safe transportation and meeting strict regulatory demands. Trust our service to deliver competitive, globally compliant products to you.
FAQs of Vildagliptin API:
Q: How is the identity and quality of Vildagliptin API verified?
A: The identity of Vildagliptin API is verified using advanced HPLC and IR spectroscopy methods, while its purity, impurity levels, and other parameters are confirmed via Certificate of Analysis provided for each batch.Q: What competitive advantages does your Vildagliptin API offer compared to others?
A: Vildagliptin API provides breathtaking purity (99%), minimal impurities (0.2%), and is manufactured in a GMP-compliant facility, ensuring reliable, first-rate quality for pharmaceutical applications.Q: Where is your Vildagliptin API exported and how is it shipped?
A: Our Vildagliptin API is exported globally, including to North America, Europe, and Asia, and is shipped in secure HDPE drums with double polyethylene bags to ensure safe transportation and storage.Q: What is the process for obtaining a sample of Vildagliptin API?
A: You may request a sample free of charge; simply contact us with your details and our team will arrange prompt processing and shipping for evaluation.Q: When should Vildagliptin API be stored and how long can it be used?
A: Vildagliptin API should be stored in a cool, dry place, protected from light. Its shelf life is 2 years from the date of manufacture when stored correctly.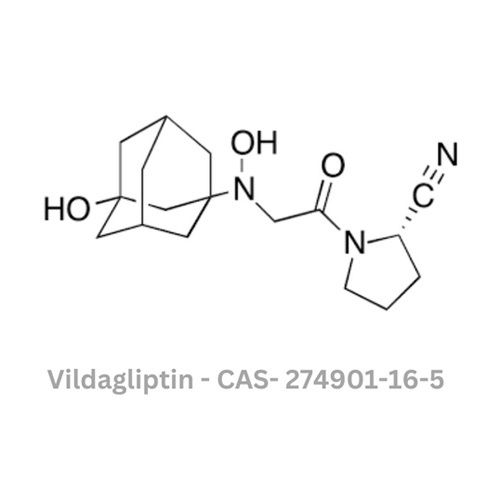

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
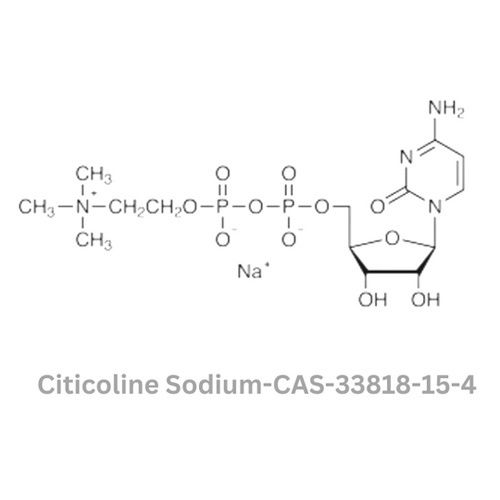
Citicoline Sodium
Minimum Order Quantity : 100 Kilograms
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade
Physical Form : Powder
Storage : Other, Store in a cool, dry place away from light and moisture
Type : Other, Active Pharmaceutical Ingredient (API)
Ursodeoxycholic Acid
Minimum Order Quantity : 100 Kilograms
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Storage : Other, Store in a cool, dry place in tightly closed containers
Type : Other, Active Pharmaceutical Ingredient
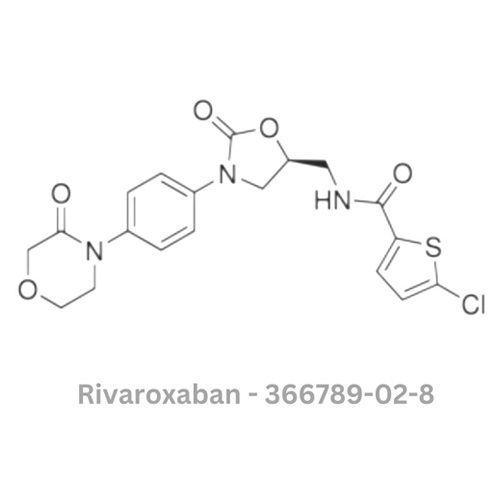
Rivaroxaban API
Minimum Order Quantity : 100 Pieces
Smell : Other, Odourless
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Storage : Other, Store in a cool, dry, wellventilated area, away from direct sunlight
Type : Other, Active Pharmaceutical Ingredient (API)
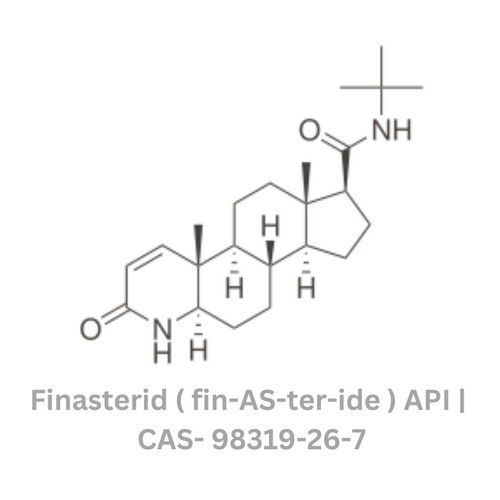
Finasterid - fin - AS - ter - ide
Minimum Order Quantity : 100 Kilograms
Smell : Stimulus
Grade : Industrial Grade
Physical Form : Powder
Storage : Room Temperature
Type : Other, Finasterid fin AS ter ide


 Send Inquiry
Send Inquiry