Bilastine API
Bilastine API Specification
- HS Code
- 29420090
- Molecular Formula
- C28H37N3O3
- Color
- White
- Molecular Weight
- 463.61 g/mol
- Particle Size
- D90 < 100 microns
- Ph Level
- 6.0-7.5 (1% aqueous solution)
- Solubility
- Freely soluble in methanol, slightly soluble in water
- Storage
- Store below 25C in a dry, well-ventilated area
- Loss on Drying
- Max 0.5%
- Smell
- Odorless
- Boiling point
- Not applicable (decomposes)
- Melting Point
- Approximately 187-189C
- Shelf Life
- 3 years from date of manufacture
- Poisonous
- Non-poisonous under normal usage
- Structural Formula
- Available on request
- Heavy Metal (%)
- 0.001%
- Taste
- Bitter
- Medicine Name
- Bilastine
- Chemical Name
- Bilastine
- CAS No
- 202825-46-5
- Type
- API (Active Pharmaceutical Ingredient)
- Grade
- Pharmaceutical Grade
- Usage
- Antihistamine, used in the treatment of allergic rhinoconjunctivitis and urticaria
- Purity(%)
- >=99%
- Appearance
- White to off-white powder
- Physical Form
- Solid
- Related Substances
- Total impurities 0.5%
- Residual Solvents
- Complies with ICH guidelines
- Transport Information
- Non-hazardous, transport under ambient conditions
- Microbial Limits
- Complies with pharmacopoeial standards
- Identification
- By HPLC and IR spectroscopy
- Assay
- 99%
- Manufacturing Capacity
- As per demand
- Packing Type
- HDPE drums
Bilastine API Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Bilastine API
Capitalise on the opportunity to order Bilastine API, manufactured to impeccable pharmaceutical standards and available at the best price from a trusted Indian exporter, manufacturer, and supplier. This dazzling antihistamine is extensively used in the treatment of allergic rhinoconjunctivitis and urticaria, offering optimum efficacy and purity (assay 99%). Residual solvents comply with ICH guidelines, while microbial limits and related substances are meticulously controlled. Packed securely in HDPE drums, its non-hazardous nature ensures hassle-free logistics. Make your next procurement seamless-order Bilastine API now for optimum pharmaceutical formulations.
Extensive Application and Usage of Bilastine API
Bilastine API is widely used by pharmaceutical companies, medical researchers, and healthcare professionals for its antihistaminic properties. The primary site of application is oral dosage forms, and its area of application broadly covers allergic rhinoconjunctivitis and chronic urticaria. Thanks to its high efficacy and targeted action, the API is valued in both hospital and outpatient settings, making it a preferred choice in allergy management protocols globally.
Domestic and International Market Presence of Bilastine API
Bilastine API commands a robust domestic market reputation in India, with widespread logistics networks ensuring prompt dispatching and delivery. Free samples are available to facilitate quality assessment. Its main export markets span Europe, Asia, Africa, and the Americas, driven by high market value and impeccable regulatory compliance standards. Reliable logistics partners streamline international shipping, upholding product quality and timely fulfillment worldwide.
Extensive Application and Usage of Bilastine API
Bilastine API is widely used by pharmaceutical companies, medical researchers, and healthcare professionals for its antihistaminic properties. The primary site of application is oral dosage forms, and its area of application broadly covers allergic rhinoconjunctivitis and chronic urticaria. Thanks to its high efficacy and targeted action, the API is valued in both hospital and outpatient settings, making it a preferred choice in allergy management protocols globally.
Domestic and International Market Presence of Bilastine API
Bilastine API commands a robust domestic market reputation in India, with widespread logistics networks ensuring prompt dispatching and delivery. Free samples are available to facilitate quality assessment. Its main export markets span Europe, Asia, Africa, and the Americas, driven by high market value and impeccable regulatory compliance standards. Reliable logistics partners streamline international shipping, upholding product quality and timely fulfillment worldwide.
FAQ's of Bilastine API:
Q: How should Bilastine API be stored for optimum shelf life?
A: Bilastine API should be stored below 25C in a dry, well-ventilated area to maintain its quality and extend its shelf life up to three years from the date of manufacture.Q: What is the process for obtaining a sample of Bilastine API?
A: Interested parties can request a sample through the supplier or manufacturer's sales team. Samples are made available to qualified clients for quality and compatibility evaluation before placing large orders.Q: Where can Bilastine API be applied in pharmaceutical formulations?
A: Bilastine API is most commonly applied in the formulation of oral tablets or syrups targeted at managing allergic symptoms such as those seen in chronic urticaria and rhinoconjunctivitis.Q: What are the benefits of using Bilastine API in medicines?
A: Bilastine API offers high purity, low impurity content, and complies with internationally recognized quality standards, ensuring effective antihistaminic action and consistent therapeutic outcomes.Q: How does Bilastine API ensure safety in transport and handling?
A: Bilastine API is classified as non-hazardous and is supplied in secure HDPE drums, enabling it to be transported under normal ambient conditions with minimal risk.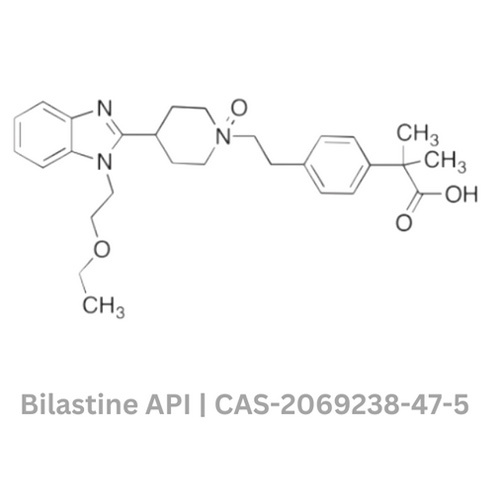
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Active Pharmaceutical Ingredients Category
Salbutamol Sulphate
Smell : Other, Odorless
Solubility : Freely soluble in water
Storage : Other, Store in cool, dry place, protect from light
Grade : Other, Pharma Grade
Physical Form : Powder
Citicoline Sodium API
Minimum Order Quantity : 100 Kilograms
Smell : Stimulus
Solubility : Yes
Storage : Room Temperature
Grade : Industrial Grade
Physical Form : Powder
Diphenhydramine HCL API
Minimum Order Quantity : 100 Kilograms
Smell : Stimulus
Solubility : Yes
Storage : Room Temperature
Grade : Industrial Grade
Physical Form : Powder
Hyoscine Butylbromide API
Minimum Order Quantity : 100 Kilograms
Smell : Stimulus
Solubility : Yes
Storage : Room Temperature
Grade : Industrial Grade
Physical Form : Powder
We accept only bulk quantity orders with more than 25 Kgs.


 Send Inquiry
Send Inquiry