Itraconazole Api
Itraconazole Api Specification
- Melting Point
- 166 - 170C
- Loss on Drying
- 0.5%
- Color
- White to Off-White
- Particle Size
- D90 < 20 m
- Poisonous
- Non-poisonous under recommended usage
- Molecular Formula
- C35H38Cl2N8O4
- Shelf Life
- 5 Years
- EINECS No
- 283-934-6
- Smell
- Odorless
- Storage
- Store in cool, dry, and well-ventilated area
- Structural Formula
- Available on request
- Heavy Metal (%)
- 0.001%
- Molecular Weight
- 705.64 g/mol
- HS Code
- 29349900
- Solubility
- Slightly soluble in Water, Soluble in Dichloromethane
- Medicine Name
- Itraconazole API
- Chemical Name
- Itraconazole
- CAS No
- 84625-61-6
- Type
- Pharmaceutical Raw Material
- Grade
- Medicine Grade
- Usage
- Antifungal Agent
- Purity(%)
- 99% Min
- Appearance
- White to Off-White Crystalline Powder
- Physical Form
- Powder
- Specific Rotation
- +21 to +25
- Residual Solvents
- Complies with ICH guidelines
- Microbial Limit
- Complies with pharmacopeial standards
- Certificate of Analysis
- Available with each batch
- Identification
- Passes test per USP standard
- Assay
- 99% - 101%
- Regulatory Documentation
- DMF available
- Related Substances
- Total Impurities 1.0%
- Polymorphic Form
- Form I
- Packaging
- HDPE drums or as per customer specification
Itraconazole Api Trade Information
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Itraconazole Api
Take advantage of our limited-time offer on the famed Itraconazole API, an uncommon antifungal pharmaceutical raw material acclaimed for its remarkable purity (99-101%) and compliance with USP standards. Each order is meticulously packaged in HDPE drums or custom containers and comes issued with a Certificate of Analysis, guaranteeing quality and regulatory assurance (DMF available). Manufactured in India by a reputed exporter, this product exhibits a white to off-white crystalline appearance, low impurities (1.0%), and impressive stability. Secure your batch now before this exclusive offer expires.
Versatile Antifungal Solution: Application and Usage
Itraconazole API is broadly utilized as an effective antifungal agent in the formulation of oral medications. Its primary surface of application is within pharmaceutical preparations targeting dermatological and systemic fungal infections. Unmatched for its efficacy against a diverse spectrum of fungal pathogens, Itraconazole API is especially remarked for its role in treating persistent infections. The powder form allows seamless blending into various medicinal compositions, ensuring optimal bioavailability and therapeutic impact in the finished dosage forms.
Global Markets and Seamless Supply: Itraconazole API Trade Solutions
Our Itraconazole API enjoys demand in famed export markets such as Europe, Asia, Latin America, and North America. We ensure prompt drop-off following a confirmed purchase order, accepting a variety of payment terms for client convenience. With a robust supply ability, we cater to both bulk and small outlay requirements, supporting pharmaceutical manufacturers with consistent and reliable product shipments worldwide. Trust our seamless process for your procurement needs.
Versatile Antifungal Solution: Application and Usage
Itraconazole API is broadly utilized as an effective antifungal agent in the formulation of oral medications. Its primary surface of application is within pharmaceutical preparations targeting dermatological and systemic fungal infections. Unmatched for its efficacy against a diverse spectrum of fungal pathogens, Itraconazole API is especially remarked for its role in treating persistent infections. The powder form allows seamless blending into various medicinal compositions, ensuring optimal bioavailability and therapeutic impact in the finished dosage forms.
Global Markets and Seamless Supply: Itraconazole API Trade Solutions
Our Itraconazole API enjoys demand in famed export markets such as Europe, Asia, Latin America, and North America. We ensure prompt drop-off following a confirmed purchase order, accepting a variety of payment terms for client convenience. With a robust supply ability, we cater to both bulk and small outlay requirements, supporting pharmaceutical manufacturers with consistent and reliable product shipments worldwide. Trust our seamless process for your procurement needs.
FAQ's of Itraconazole Api:
Q: How is Itraconazole API typically used in pharmaceuticals?
A: Itraconazole API is primarily used to formulate antifungal medications, both oral and systemic, targeting chronic and acute fungal infections in humans.Q: What documentation is provided with an order of Itraconazole API?
A: Each batch is accompanied by a Certificate of Analysis and full regulatory documentation, including Drug Master File (DMF), ensuring compliance and transparency.Q: Where can Itraconazole API be shipped from?
A: Itraconazole API is exported from India, packaged securely to meet international shipping regulations and customer specifications.Q: What is the process for placing a purchase order for Itraconazole API?
A: To place an order, simply contact our sales team with your product requirements. We provide prompt quotations, confirm payment terms, and arrange reliable shipment upon order confirmation.Q: What are the storage and shelf life requirements for Itraconazole API?
A: Itraconazole API should be stored in a cool, dry, and well-ventilated area. When properly stored, it maintains its stability for up to five years.Q: What are the main benefits of choosing your Itraconazole API?
A: Our Itraconazole API offers exceptional purity, compliance with international standards, a proven safety profile, and reliable global logistics for timely delivery.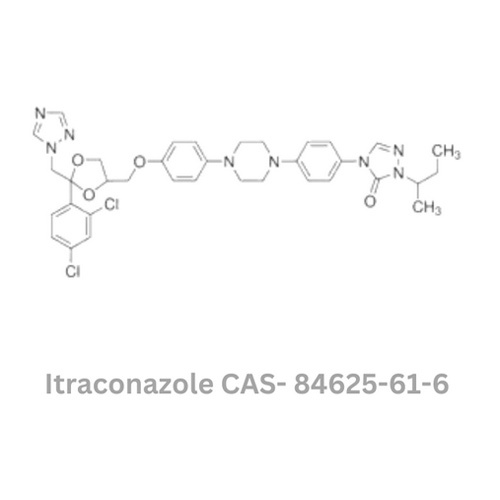
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Active Pharmaceutical Ingredients Category
Hydrochloride Monohydrate
Smell : Other, Odorless
Grade : Other, Pharmaceutical
Storage : Other, Store in cool, dry place
Physical Form : Solid
Taste : Bitter
Gliclazide API
Smell : Other, Odorless
Grade : Other, Pharmaceutical Grade
Storage : Other, Store in a cool, dry place, protected from light
Physical Form : Solid
Taste : Bitter
Citicoline Sodium API
Minimum Order Quantity : 100 Kilograms
Smell : Stimulus
Grade : Industrial Grade
Storage : Room Temperature
Physical Form : Powder
Taste : Bitter
levosalbutamol Sulphate API
Minimum Order Quantity : 100 Kilograms
Smell : Stimulus
Grade : Industrial Grade
Storage : Room Temperature
Physical Form : Powder
Taste : Bitter
We accept only bulk quantity orders with more than 25 Kgs.


 Send Inquiry
Send Inquiry