Ondansetron Hydrochloride
Ondansetron Hydrochloride Specification
- Loss on Drying
- 0.5%
- Taste
- Tasteless
- Structural Formula
- C18H19N3OHCl
- EINECS No
- 627-129-7
- Smell
- Odorless
- Melting Point
- Approximately 178-180C
- Heavy Metal (%)
- 0.001%
- Molecular Weight
- 329.82 g/mol (base), 365.83 g/mol (hydrochloride salt)
- Shelf Life
- 36 months
- Ph Level
- 4.0 - 5.0 (1% aqueous solution)
- Poisonous
- No (When used as directed under medical supervision)
- Color
- White to off-white
- Solubility
- Freely soluble in water and methanol, sparingly soluble in ethanol
- Storage
- Store in a cool, dry place, protected from light and moisture.
- HS Code
- 29339900
- Boiling point
- Not applicable (decomposes)
- Particle Size
- D90 < 100 microns
- Molecular Formula
- C18H19N3OHCl
- Medicine Name
- Ondansetron Hydrochloride
- Chemical Name
- Ondansetron Hydrochloride
- CAS No
- 99614-02-5
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- Pharmaceutical Grade
- Usage
- Used as an antiemetic to prevent nausea and vomiting associated with cancer chemotherapy, radiotherapy, and surgery.
- Purity(%)
- >99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Chloride Content
- 16.0% - 17.0%
- Packaging
- HDPE drum with double polyethylene bags
- Assay (by HPLC)
- 99.0%
- Identification (IR, UV)
- Complies
- pKa
- 7.4 (approximate)
- Residual Solvents
- Complies with ICH guidelines
- Microbial Limit
- Complies with pharmacopeial standards
- Related Substances
- 0.2%
Ondansetron Hydrochloride Trade Information
- Minimum Order Quantity
- 100 Kilograms
- Supply Ability
- 1000 Kilograms Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Ondansetron Hydrochloride
- Generic Name : Ondansetron Hydrochloride
- CAS Number : 67914-60-7
- Grade : IP/BP/USP
- Packing Type : Drum
- Therapeutic use : Antagonist, Antiemetics, Antinauseants
- Product MOQ : 25 kgs
Unmatched Purity and Compliance
Ondansetron Hydrochloride stands out with an assay of 99.0% and purity greater than 99%, ensuring exceptional quality for pharmaceutical applications. Every batch complies with ICH guidelines for residual solvents, as well as stringent pharmacopeial standards for microbial and heavy metal content, making it highly reliable for medical use.
Versatile Antiemetic Use
This API is widely used in the formulation of antiemetic medications. Ondansetron Hydrochloride helps prevent and control nausea and vomiting induced by cancer chemotherapy, radiotherapy, and surgery, improving patient quality of life and treatment outcomes. Its tasteless and odorless profile ensures suitability for a range of oral and injectable formulations.
Safety, Packaging, and Storage
Ondansetron Hydrochloride is securely packaged in HDPE drums with double polyethylene bags, preserving its integrity during storage and transport. It should be stored in a cool, dry place, shielded from light and moisture, and offers a shelf life of 36 months. Compliance with storage recommendations ensures sustained potency and safety.
FAQ's of Ondansetron Hydrochloride:
Q: How is Ondansetron Hydrochloride typically used in medical settings?
A: Ondansetron Hydrochloride is used as an active ingredient in antiemetic medications to prevent nausea and vomiting caused by cancer chemotherapy, radiotherapy, and surgical procedures. It is formulated into oral or injectable dosage forms, prescribed and administered under medical supervision.Q: What measures ensure the quality and safety of Ondansetron Hydrochloride?
A: This API undergoes stringent testing for purity, with an HPLC assay of 99.0%, and compliance with ICH guidelines for residual solvents and pharmacopeial standards for microbial and heavy metal content. Its identification is confirmed by IR and UV, and each batch is securely packaged to maintain product integrity.Q: When should Ondansetron Hydrochloride be stored to maintain optimal quality?
A: It should always be stored in a cool, dry environment, protected from moisture and direct sunlight. Proper storage conditions help preserve its physical and chemical stability, ensuring a 36-month shelf life from the date of manufacture.Q: Where is Ondansetron Hydrochloride typically manufactured and supplied from?
A: Ondansetron Hydrochloride is manufactured, exported, and supplied widely from India. The country is a leading hub for pharmaceutical-grade APIs, ensuring adherence to international quality and regulatory requirements.Q: What is the solubility profile of Ondansetron Hydrochloride?
A: Ondansetron Hydrochloride is freely soluble in water and methanol, but sparingly soluble in ethanol, allowing flexibility in formulation for both oral and injectable preparations.Q: What are the main benefits of using Ondansetron Hydrochloride as an antiemetic?
A: Its high purity, excellent compliance with safety standards, and proven efficacy in controlling nausea and vomiting make Ondansetron Hydrochloride a preferred choice for pharmaceutical manufacturers. Patients benefit from improved treatment tolerance during chemotherapy, radiotherapy, or surgery.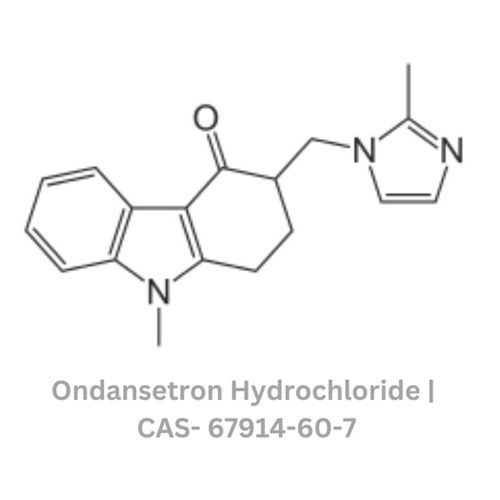

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
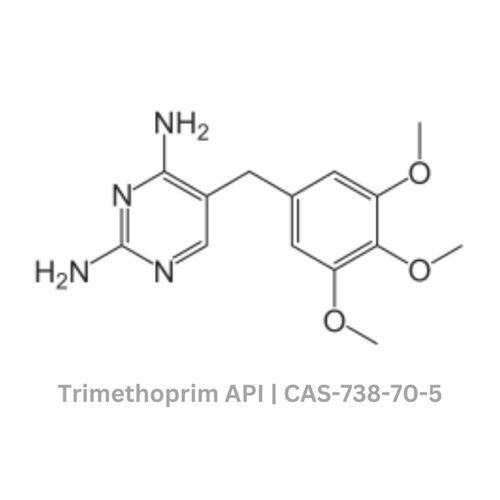
Trimethoprim API
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Taste : Bitter
Smell : Other, Odorless
Type : Other, Active Pharmaceutical Ingredient
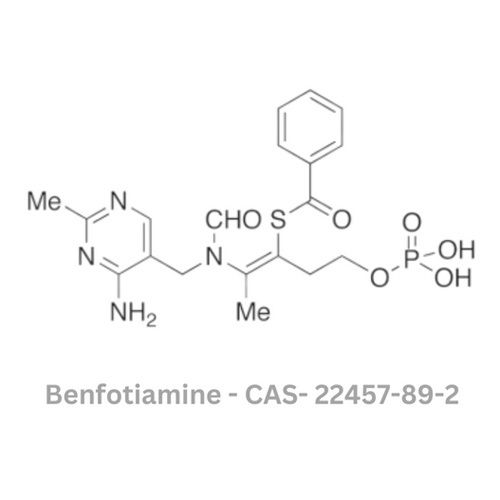
Benfotiamine API
Minimum Order Quantity : 120 Kilograms
Grade : Other, Pharmaceutical Grade
Physical Form : Solid
Taste : Other , Slightly Bitter
Smell : Other, Odorless
Type : Other, Active Pharmaceutical Ingredient
Ketorolac Tromethamine API
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Physical Form : Powder
Taste : Bitter
Smell : Stimulus
Type : Other, Ketorolac Tromethamine API
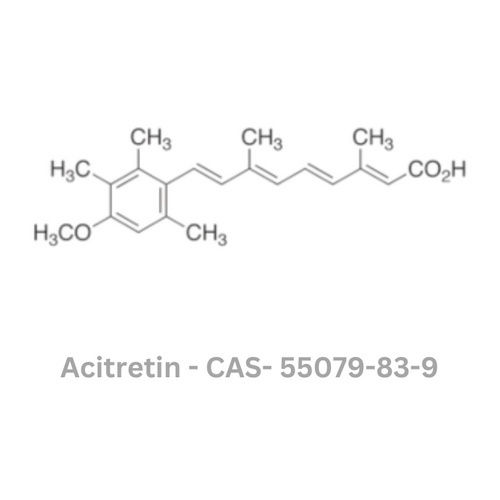
Acitretin API
Minimum Order Quantity : 100 Kilograms
Grade : Industrial Grade
Physical Form : Powder
Taste : Odorless
Smell : Sharp
Type : Other, Acitretin API


 Send Inquiry
Send Inquiry