Aripiprazole API
Aripiprazole API Specification
- Loss on Drying
- 0.5%
- Storage
- Store in a cool, dry place, protected from light
- Solubility
- Slightly soluble in water, soluble in methanol and ethanol
- Shelf Life
- 2 years if properly stored
- Structural Formula
- Available on request
- Poisonous
- NO
- Heavy Metal (%)
- 10 ppm
- Molecular Weight
- 448.39 g/mol
- Molecular Formula
- C23H27Cl2N3O2
- HS Code
- 29339900
- Color
- White
- Particle Size
- As per specifications, available upon request
- Smell
- Odourless
- Melting Point
- 139-141C
- Medicine Name
- Aripiprazole API
- Chemical Name
- 7-{4-[4-(2,3-dichlorophenyl)-1-piperazinyl]butyloxy}-3,4-dihydro-2(1H)-quinolinone
- CAS No
- 129722-12-9
- Type
- Active Pharmaceutical Ingredient
- Grade
- Pharmaceutical Grade
- Usage
- Used in treatment of schizophrenia and bipolar disorder
- Purity(%)
- 99%
- Appearance
- White or almost white powder
- Physical Form
- Solid
- Chloride Content
- 0.014%
- Microbial Limits
- Complies with pharmacopeial standards
- Related Substances
- 0.2%
- Packing
- HDPE drum with double polyethylene bag
- Identification
- By IR and HPLC
- Regulatory Status
- DMF available
- Assay (by HPLC)
- >= 99.0%
- Residual Solvents
- Complies with ICH guidelines
Aripiprazole API Trade Information
- Minimum Order Quantity
- 100 Kilograms
- Supply Ability
- 1000 Kilograms Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Aripiprazole API
Versatile Usage and Application Method of Aripiprazole API
Aripiprazole API is designed primarily for pharmaceutical manufacturers formulating medications to treat schizophrenia and bipolar disorder. Its surface of application includes blending within solid dosage forms like tablets or capsules. The application method involves precise weighing, dissolving, or direct mixing, adhering strictly to formulation guidelines. The API's excellent solubility in methanol and ethanol makes it suitable for complex pharmaceutical preparations, ensuring optimal efficacy in various medicinal products. Detailed particle size information is available upon request to tailor formulation processes.
Efficient Delivery and Sample Availability for Aripiprazole API
On receiving your purchase order, we prioritize an organized handover process, culminating in rapid dispatching of Aripiprazole API. Delivery timelines within our main domestic market, India, are optimized for prompt and reliable supply. As a commitment to quality assurance, sample quantities are available for potential collaborators, allowing comprehensive evaluation before committing to bulk orders. Our robust packaging process in HDPE drums with double polyethylene liners guarantees the API's integrity throughout transit and storage.
FAQ's of Aripiprazole API:
Q: How is the quality of Aripiprazole API ensured during manufacture?
A: The quality of Aripiprazole API is maintained through strict compliance with ICH guidelines, pharmacopeial standards, validated HPLC assays, and precise control of related substances, ensuring a consistent, first-class product.Q: What is the typical usage of Aripiprazole API?
A: Aripiprazole API is primarily used in the pharmaceutical industry for formulating medications aimed at treating schizophrenia and bipolar disorder.Q: When should Aripiprazole API be stored after opening?
A: Immediately after opening, Aripiprazole API should be stored in a cool, dry place, protected from light, and in its original packaging to preserve its 2-year shelf life.Q: Where can I obtain the structural formula or particle size data for Aripiprazole API?
A: Both the structural formula and particle size data for Aripiprazole API are available upon request from the manufacturer or supplier.Q: What is the process for receiving a sample of Aripiprazole API?
A: You can request a sample alongside your inquiry or purchase order, and our team will arrange dispatching a sample for your evaluation.Q: How is the product dispatched and what packaging is used?
A: Aripiprazole API is safely packed inside HDPE drums with a double polyethylene liner and dispatched promptly after confirming the purchase order.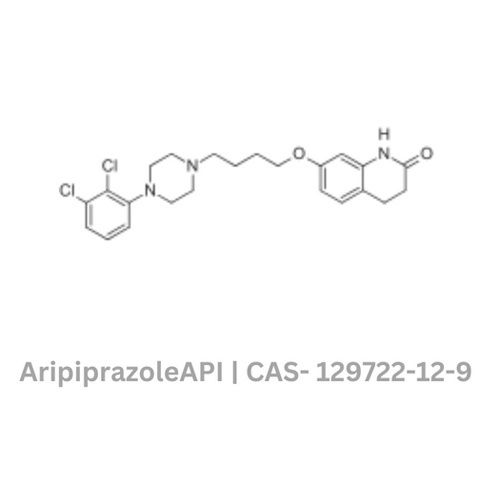

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Active Pharmaceutical Ingredients Category
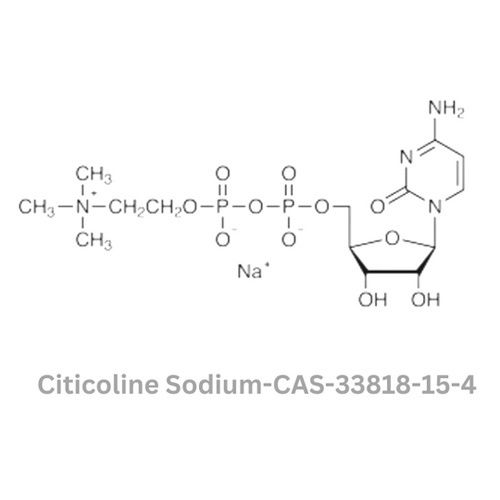
Citicoline Sodium
Minimum Order Quantity : 100 Kilograms
Usage : Used in the treatment of stroke, cognitive impairment, and neurological disorders
Grade : Other, Pharmaceutical Grade
Color : Other, White to offwhite
Type : Other, Active Pharmaceutical Ingredient (API)
Storage : Other, Store in a cool, dry place away from light and moisture
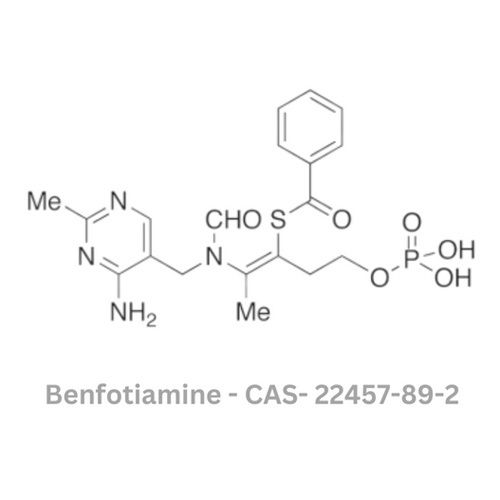
Benfotiamine API
Minimum Order Quantity : 120 Kilograms
Usage : Used as a vitamin supplement, especially for diabetic neuropathy and other vitamin B1 deficiency conditions
Grade : Other, Pharmaceutical Grade
Color : Other, White or almost white
Type : Other, Active Pharmaceutical Ingredient
Storage : Other, Store in cool, dry and wellventilated area, away from moisture and direct sunlight
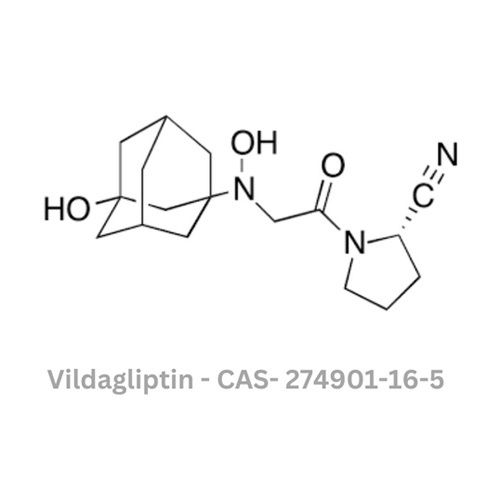
Vildagliptin API
Usage : Antidiabetic agent, used in the treatment of type 2 diabetes mellitus
Grade : Other, Pharmaceutical Grade
Color : Other, White to offwhite
Type : Other, Active Pharmaceutical Ingredient (API)
Storage : Other, Store in a cool, dry place, protected from light
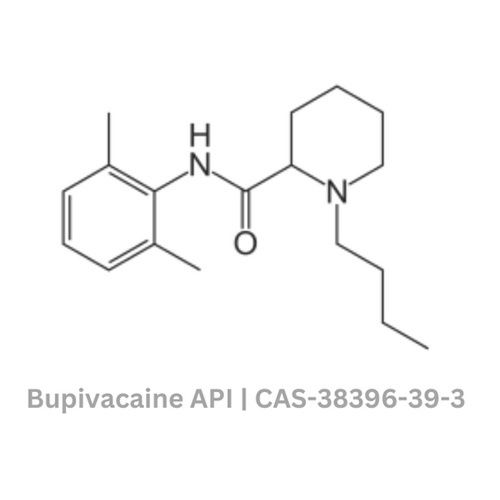
Bupivacaine API
Minimum Order Quantity : 100 Kilograms
Usage : Commercial
Grade : Industrial Grade
Color : White
Type : Other, Bupivacaine API
Storage : Room Temperature


 Send Inquiry
Send Inquiry